| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://www.neurores.org |
Original Article
Volume 11, Number 1-2, April 2021, pages 5-13
Perioperative Stroke Impact on Morbidity and In-Hospital Mortality in Non-Cardiovascular Non-Neurological Surgeries: State Inpatient Database Study
Samir S. Al-Ghazawia, Khaled A. Al-Robaidib, Ashutosh P. Jadhavc, Qasem Al Shaerd, Nada Alrifaie, Parthasarathy D. Thirumalaf, g, h
aDepartment of Neurology, Memorial Healthcare System, Hollywood, FL, USA
bDepartment of Pediatric Neurology, University of at Alabama Birmingham, Birmingham, AL, USA
cDepartment of Neurosurgery, Barrow Neurological Institute, Phoenix, AZ, USA
dDepartment of Neurology, Emory University, Atlanta, GA, USA
eDepartment of Internal Medicine, Allegheny General Hospital, Pittsburgh, PA, USA
fDepartment of Neurology, University of Pittsburgh, Pittsburgh, PA, USA
gDepartment of Neurological Surgery, University of Pittsburgh, Pittsburgh, PA, USA
hCorresponding Author: Parthasarathy D. Thirumala, Center for Clinical Neurophysiology, Department of Neurological Surgery, UPMC Presbyterian, Suite B-400, 200 Lothrop Street, Pittsburgh, PA 15213, USA
Manuscript submitted February 25, 2021, accepted April 16, 2021, published online April 24, 2021
Short title: SID Study on Perioperative Stroke Impact
doi: https://doi.org/10.14740/jnr653
| Abstract | ▴Top |
Background: Perioperative stroke, which occurs within 30 days following surgery, can increase morbidity and mortality. The prior might reduce surgical interventions’ clinical effectiveness, carrying significant impact on patients, their families, and healthcare resources utilization. We examined the impact of perioperative stroke on morbidity and mortality in patients undergoing non-cardiovascular non-neurological surgeries.
Methods: We performed a retrospective cohort study utilizing State Inpatient Database (SID) available from the Health Cost Utilization Project (HCUP) between 2008 and 2011. The databases above can distinguish between previous and new strokes by providing the “present on admission” variable. We used multivariable logistic regression to assess stroke as an independent predictor of morbidity and mortality after adjusting for other covariables.
Results: From the SID, a total of 1,206,057 inpatient discharge records were identified. The incidence of perioperative stroke, in-hospital mortality, and morbidity was 0.14%, 0.54%, and 8.1%. Multivariate logistic regression showed that perioperative stroke is an independent predictor of morbidity (odds ratio (OR) = 5.06, 95% confidence interval (CI) = 4.54 - 5.64, P < 0.001) and in-hospital mortality (OR = 7.26, 95% CI = 6.26 - 8.41, P < 0.001). Other independent predictors were identified, but they showed a weaker association with in-hospital mortality than perioperative stroke, including cardiovascular complications (OR = 4.33, CI = 3.93 - 4.77, P < 0.001).
Conclusions: Perioperative stroke is a significant independent risk factor for morbidity and in-hospital mortality following non-cardiovascular, non-neurological surgeries. Patients who developed perioperative stroke had a higher risk of death than perioperative cardiovascular and pulmonary complications.
Keywords: Perioperative; Stroke; Morbidity; In-hospital mortality; Surgery
| Introduction | ▴Top |
Morbidity and mortality after non-cardiovascular non-neurological surgeries can significantly impact patients, their families, and the utilization of healthcare resources [1]. Perioperative stroke, defined as a new stroke developing within 30 days following surgery, is a recognized complication, with reported incidences ranging from 0.2% in hip replacement to 0.7% in hemicolectomy [2]. The impact of perioperative stroke on morbidity and mortality is well described in cardiovascular and neurological surgeries, with a high incidence ranging from 1.9% to 9.7% [1, 3, 4].
Previous studies regarding perioperative stroke in non-cardiovascular non-neurological surgeries have shown that patients with perioperative stroke had a longer hospital stay and higher mortality. However, these studies had a smaller sample size and focused on a limited set of surgical procedures, which did not provide a comprehensive overview of perioperative stroke’s actual impact on the global burden of surgical diseases [1, 2, 5, 6].
In the USA alone, a significant increase (14-47%) in demand for surgical services is expected over the coming years [1]. The health care system’s performance is assessed by both patients’ short-term and long-term outcomes. Short-term outcomes include in-hospital mortality and complications, and long-term outcomes incorporate long-term disability and neurological deficits [7, 8]. The rise of perioperative morbidity and in-hospital mortality can reduce the effectiveness of surgical procedures [9]. Improving patient outcomes requires predicting populations at elevated risk of perioperative complications, including perioperative stroke [10].
As the population ages, the number of comorbidities increases, which can directly impact perioperative stroke incidence [11]. Comorbidities may include atrial fibrillation, hypertension, diabetes mellitus, hypercholesterolemia, renal impairment, previous history of myocardial infarction, and previous stroke history [12, 13]. Therefore, understanding the relationship between patient risk factors, surgical procedure risk factors, and perioperative outcomes is essential for a meaningful predictive model [9, 14].
We aim to perform a retrospective study to assess perioperative stroke’s impact as an independent predictive factor for morbidity and in-hospital mortality in non-cardiovascular, non-neurological surgeries. We hypothesize that perioperative stroke increases morbidity and in-hospital mortality. This study will help in understanding the impact of perioperative stroke and help in screening patients before surgery and improve patient outcomes by utilizing intraoperative neuromonitoring (IONM) as intraoperative electroencephalogram (EEG) and somatosensory evoked potentials (SSEP) [15, 16]. As the latter can help detect stroke and allow for aggressive interventional or medical management after the surgical procedures [17-19].
| Materials and Methods | ▴Top |
Study design and data source
This study is a retrospective cohort study. The Institutional Review Board (IRB) approval was exempted because the database we used does not contain patients’ identifiers; and the study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration. Data were derived from the State Inpatient Database (SID), part of the Healthcare Cost and Utilization Project (HCUP). The SID represents about 97% of all US community hospital discharges. It contains a core set of clinical and nonclinical information on patients, including individuals covered by Medicare, Medicaid, or private insurance, as well as those who are uninsured. SID applies more than 100 clinical and nonclinical variables, including principal and secondary diagnoses and procedures, patient demographic characteristics such as sex, age, race, total charges, expected payer, length of stay (LOS), admission, and discharge status. Data were collected from 2008 to 2011. California SID has present on admission (POA), which helps differentiate whether a stroke developed as a complication from surgery versus stroke from a miscellaneous etiology prior to admission [20].
Patient selection
Patients included in this study were patients aged 18 years old and above. Surgical procedures were identified using their corresponding International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes. For our study, we selected the most common 10 surgical procedures performed from the HCUP list “Most Common Operations during Inpatient Stays” [21]. Surgeries included in the study were hemicolectomy, appendectomy, cholecystectomy, hip replacement, knee replacement, amputation of the lower extremity, laminectomy, spine fusion, and hysterectomy. We excluded patients who underwent cardiac, major vascular, neonatal, high-risk neurological procedures, obstetric procedures, and diagnostic procedures.
Covariables and study outcomes
We identified risk factors for perioperative stroke from published review articles, case series, and clinical plausibility. Patient demographic covariates included age groups (< 65, 65 - 74, 75 - 84, ≥ 85 years), gender, and race. Clinical covariates included admission type (elective vs. emergency), Elixhauser comorbidity variables, and van Walraven score (< 5, 5 - 14, > 14). Perioperative stroke was identified using the corresponding ICD-9-CM codes, including ischemic, hemorrhagic, and iatrogenic stroke. Iatrogenic stroke (ICD-9-CM 997.02) is a billing code used to describe any stroke in the perioperative period in SID (Supplementary Material 1, www.neurores.org) [2]. Mortality was defined as death during the hospital stay. Morbidity was defined as the prolonged LOS exceeding 14 days from surgery, or developing cardiovascular complications, pulmonary complications, gastrointestinal complications, post-operative infection, wound complications, and renal complications [22]. Our primary outcome of interest was in-hospital mortality, and our secondary outcome was morbidity.
Statistical analysis
Continuous data were represented as means and standard deviation (SD). Categorical data were presented as percentages. Independent sample t-test was used for assessing the relationship between continuous variables and Chi-square for categorical variables. Variables that were significant in the bivariate analysis were included in the final regression model. Multivariate logistic regression model was conducted to estimate the adjusted odds of morbidity and in-hospital mortality. A second multivariate regression analysis was conducted for each subgroup of morbidity to assess perioperative stroke’s impact on other perioperative complications. The Hosmer-Lemeshow test was used to determine goodness-of-fit. Database construction was done using SAS statistical analysis program. Statistical analysis was carried utilizing IBM SPSS Statistics version 23® (Armonk, NY).
| Results | ▴Top |
From the California SID dataset, a total of 1,206,057 inpatient discharge records spanning the years 2008 - 2011 were identified (Table 1). The mean age for subjects without stroke at the time of admission was 57.9 years (SD = 19), as compared to 74.3 years (SD = 13.8) for subjects with stroke. Subjects in the sample were predominately female (63.4%). Our sample included patients who underwent the following surgeries: knee replacement (17.45%), hysterectomy (14.3%), cholecystectomy (13.5%), hip replacement (12.3%), spine fusion (10.9%), appendectomy (10.2%), treatment of fracture or dislocation of lower extremity (7.7%), laminectomy (6.7%), amputation of lower extremity (3.14%), and hemicolectomy (3.11%).
 Click to view | Table 1. Patients Characteristics |
The largest age group in our sample was those under 65 years of age (59.7 %). The mean age of males was 58.7 years (SD = 18.7), the mean age of females was 58.4 years (SD = 18.6). Whites composed the majority of subjects (60.6%), followed by Hispanics (18.5%) and Blacks (5%). Surgical procedures were predominately elective cases (66.1%) and had an average LOS of 4.04 days (SD = 5.2). The mean van Walraven score was found to be 1.32 (SD = 4.9), with most (82 %) of subjects scoring < 5 on a scale ranging from -17 to 53 in the studied sample.
Incidence of stroke
Perioperative stroke was found to have an overall incidence rate of 0.14% (n = 1,739) in the whole sample. Rates of perioperative stroke increased progressively with age in those over 85 years of age (0.60%, n = 457), exhibiting a rate 12 times higher than those under 65 years of age (0.05%, n = 364, P < 0.001). Males had a risk of stroke (0.17%) greater than females (0.14%, P < 0.001). The three procedures with the highest observed risk of perioperative stroke were hemicolectomy (0.50%, n = 189), amputation of lower extremity (0.44%, n = 167), treatment of fracture or dislocation of lower extremity (0.41%, n = 382). The remaining procedures all had rates of stroke at or below 0.2%, with the risk of stroke in appendectomy being the lowest (0.02%, n = 27).
Incidence of mortality
Overall in-hospital mortality was 0.56% (n = 6,810), with those dying (75.26 years, SD = 13.9) being approximately 18 years older than those who survived (57.81 years, SD = 19.07, P < 0.001). Furthermore, 31.5% of the total in-hospital mortality was observed in patients who were 85 years old and above, although this population accounted for only 6.8% (n = 81,141) of the studied sample. Mortality rates were highest in patients who underwent hemicolectomy (4.2%), followed by amputation of lower extremity (2.79%) (Supplementary Material 2, www.neurores.org). The risk of mortality in an emergency admission was seven times higher than in an elective admission (odds ratio (OR) = 7.04, 95% confidence interval (CI) = 6.63 - 7.48, P < 0.001). On bivariate analysis, rates of in-hospital mortality were the highest in patients with stroke (18.9%, OR = 43.18, 95% CI = 38.20 - 48.81) when compared to cardiovascular (7.4%, OR = 15.74, 95% CI = 14.54 - 17.04, P < 0.001) and pulmonary complications (10.2%, OR = 24.39, 95% CI = 22.90 - 25.98, P < 0.001) (Fig. 1).
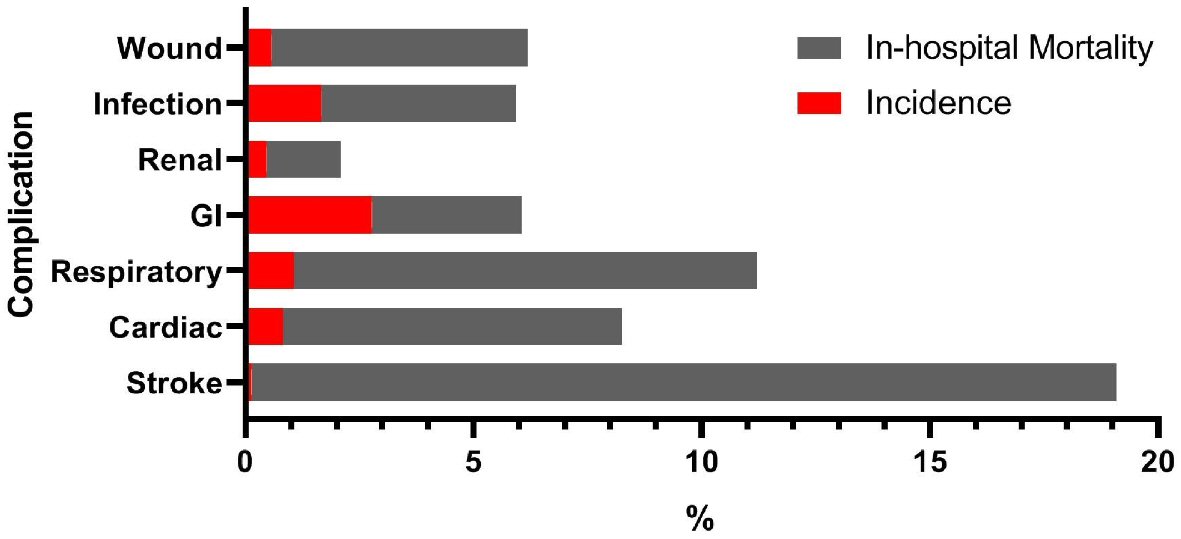 Click for large image | Figure 1. The incidence of perioperative complication and its corresponding in-hospital mortality. GI: gastrointestinal. |
Incidence of morbidity
The overall incidence of morbidity was 8.1% (n = 98,106). The morbidity was higher among patients older than 85 years (15.6%, n = 12,671) (Supplementary Material 2, www.neurores.org). Males exhibited higher morbidity than females (10.4% vs. 7.3%). Subjects who underwent hemicolectomy exhibited the highest portion of post-operative infection followed by treatment of fracture of the lower extremity with the incidence of 4.7%, 4.2%, respectively. Subjects who underwent hemicolectomy exhibited the highest portion of wound complications followed by amputation of lower extremity with incidence of 5.4%, 1.21%, respectively. Subjects who underwent hip replacement exhibited the highest portion of gastrointestinal complications followed by hysterectomy with incidence of 17.4%, 14.3%, respectively. Subjects who underwent hemicolectomy exhibited the highest portion of renal complications followed by laminectomy and spine fusion with incidence of 1.1%, 0.6%, respectively.
Multivariate analysis
A multivariate logistical regression analysis was conducted to test for the association of perioperative stroke with morbidity and mortality while adjusting for age, sex, race, procedure type, admission type, and comorbidities (Table 2). Perioperative stroke was found to be a significant risk factor for both morbidity (OR =5.06, CI = 4.54 - 5.64, P < 0.001) and in-hospital mortality (OR = 7.26, 95% CI = 6.26 - 8.41, P < 0.001) (Fig. 2a). A separate multivariate logistical regression for morbidity subgroups was carried to assess the impact of perioperative stroke on perioperative complications (Supplementary Material 3, www.neurores.org). It showed that perioperative stroke remained a significant independent predictor for prolonged LOS (OR = 7.59, 95% CI = 6.73 - 8.57, P < 0.001), cardiovascular complications (OR = 3.40, 95% CI = 2.80 - 4.12, P < 0.001), respiratory complications (OR = 3.10, 95% CI = 2.68 - 3.73, P < 0.001), post-operative infection (OR = 2.81, 95% CI = 2.43 - 3.25, P < 0.001), wound complications (OR = 2.28, 95% CI = 1.73 - 3.00, P < 0.001), renal complications ( OR = 1.72, 95% CI = 1.16 - 2.56, P < 0.001), and gastrointestinal complications (OR = 1.99, 95% CI = 1.67 - 2.38, P < 0.001) (Fig. 2b).
 Click to view | Table 2. Multivariable Regression Model 1: Morbidity and Mortality |
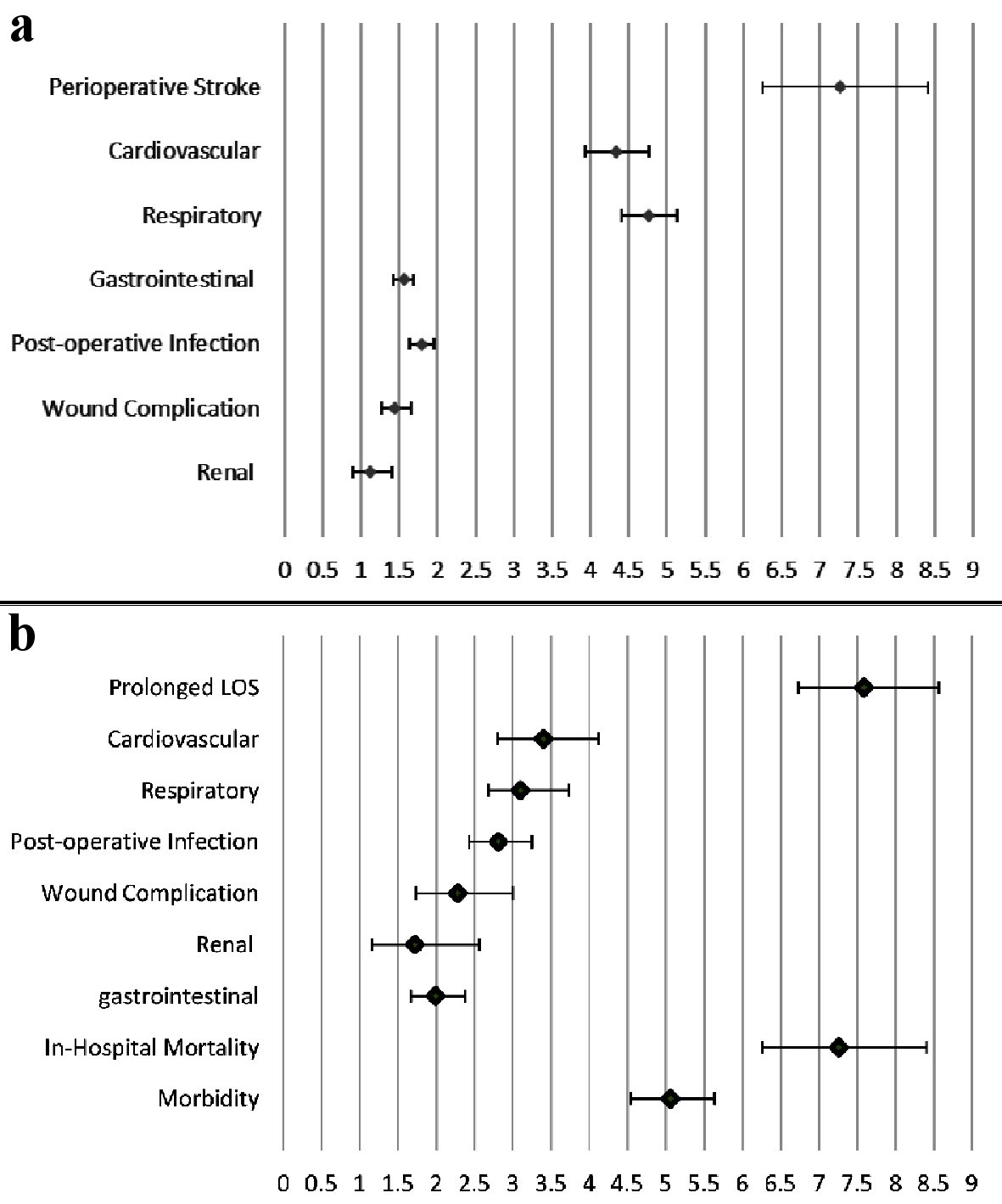 Click for large image | Figure 2. (a) The factors’ impact on in-hospital mortality demonstrated by odds ratio and 95% confidence intervals. (b) The impact of perioperative stroke on morbidity, mortality, cardiovascular, respiratory, infection, wound, renal and gastrointestinal complications demonstrated by odds ratio and 95% confidence intervals. LOS: length of stay. |
| Discussion | ▴Top |
Our results confirm that perioperative stroke is a significant risk factor for morbidity and in-hospital mortality following non-cardiovascular, non-neurological surgeries. In the SID, a retrospective cohort study of 1,206,057 patients showed an incidence of perioperative stroke ranging from 0.02% in appendectomy to 0.50% following hemicolectomy, with an overall incidence of 0.14%, which is lower than the reported incidence of stroke in papers that focused on cardiovascular surgeries which range from 1.9% to 9.7% [4]. A previous study by Bateman et al showed incidences of perioperative stroke ranging from 0.2% after appendectomy to 0.7% after hemicolectomy compared to the 0.14% overall incidence in our study [2]. One interpretation for the previous result is in the study of Bateman et al, it only included three surgeries in Nationwide Inpatient Sample (NIS). NIS data do not have a present on admission indicator, which might result in difficulty in distinguishing if the strokes occurred in the hospital or occurred before admission. The prior was reflected on the reported incidence of perioperative stroke. The study of Mashour et al showed that the overall incidence of perioperative stroke in non-cardiac non-neurological surgeries using ACS National Surgical Quality Improvement Program (NSQIP) data set is 0.1%, which is similar to our results [1].
Little attention has been given to perioperative stroke than cardiovascular complications, as the former usually has a lower incidence. However, in our study, we found that patients with perioperative stroke had a seven-fold increase in in-hospital mortality compared to the four-fold increase in in-hospital mortality from cardiovascular complications (Fig. 2a). Despite the lower incidence of perioperative stroke, its impact on in-hospital mortality was detrimental. A recent study by Smilowitz et al showed a reduction in the death rate from acute myocardial infarction among patients undergoing major non-cardiac surgery in the USA; however, in the same study, they showed that the incidence of ischemic stroke increased over time [23].
Since the Goldman index introduction, cardiovascular events following surgeries have continuously declined, mainly attributed to appropriate patient selection for the surgeries. Goldman index is a tool that weighs patient demographics, comorbidities, signs of heart failure, electrocardiographic abnormalities, general medical conditions, and the type of operation to designate an appropriate class that correlates with a specific postoperative risk for cardiac complications [24]. Therefore, the presence of a meaningful predictive model to predict the risk of perioperative stroke and possible interventions during the perioperative period could decrease the incidence of perioperative stroke. Some measures which have been shown to detect and reduce perioperative stroke include different intraoperative anesthetic techniques, pharmacologic strategies, and physiologic strategies as intraoperative neuromonitoring (IONM). IONM include intraoperative EEG and SSEP [1, 25, 26]. Combining IONM and utilizing appropriate stroke units for post-stroke care can reduce the morbidity and mortality in patients who suffer from perioperative stroke [27].
In our study, we found that female gender was associated with lower risk for morbidity (OR = 0.82, 95% CI = 0.81 - 0.83, P < 0.001) and mortality (OR = 0.76, 95% CI = 0.72 - 0.80, P < 0.001) in comparison to males. The prior was congruent with the literature as the female gender was associated with lower mortality and shorter LOS. Furthermore, stroke etiology subtypes also differ between males and females, as cardioembolic etiology was more frequent in females, while small vessel disease was more frequent in males [28].
The cardioembolic phenomenon is one of the most common etiologies of perioperative stroke. Atrial fibrillation (AF) is the most common arrhythmia in the perioperative period, and the risk for AF increases significantly in the perioperative period, especially in non-cardiac surgery. Furthermore, the outcomes for cardioembolic stroke are drastic in comparison to other stroke etiologies. Therefore, different measures can be taken to reduce AF risk, as avoiding potential triggers and treating patient and surgery-related risk factors that might precipitate AF [29, 30].
Limitations
Despite the significant benefits of our study, we do have some limitations. This study utilized the SID hospital claims database. One of the limitations of that dataset is the accuracy of some ICD-9-CM-coded diagnoses and procedures, including miscoding comorbidities [20]. The lack of ability to prospectively follow up the patients for the possibility of readmission, and complications following their discharge, limits the ability to evaluate the long-term impact of perioperative stroke.
Conclusions
Perioperative stroke is a significant independent risk factor for morbidity and in-hospital mortality following non-cardiovascular, non-neurological surgeries. Patients who develop perioperative stroke have a higher risk of death compared to perioperative cardiac complications. Patients with perioperative stroke have a higher risk of other perioperative complications, including cardiovascular, respiratory, post-operative infection, wound complications, renal, gastrointestinal complications, and prolonged hospital LOS.
| Supplementary Material | ▴Top |
Suppl 1. ICD-9 code descriptions.
Suppl 2. General characteristics and initial bivariate analysis.
Suppl 3. . Multivariable regression model 2: morbidity subgroups.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
The authors declare no conflict of interest.
Informed Consent
Informed consents were not needed because there were no patients’ identifiers in the database.
Author Contributions
Samir S. Al-Ghazawi: hypothesis, writing, data analysis, literature search, and submission. Khaled A. Al-Robaidi: data analysis, editing, and reviewing. Ashutosh P. Jadhav: reviewing and editing. Qasem Al Shaer: literature search, editing, and figure design. Nada Alrifai: reviewing and editing. Parthasarathy D. Thirumala: hypothesis, editing, supervision, and reviewing.
Data Availability
Any inquiries regarding supporting data availability of this study should be directed to the corresponding author.
| References | ▴Top |
- Mashour GA, Moore LE, Lele AV, Robicsek SA, Gelb AW. Perioperative care of patients at high risk for stroke during or after non-cardiac, non-neurologic surgery: consensus statement from the Society for Neuroscience in Anesthesiology and Critical Care*. J Neurosurg Anesthesiol. 2014;26(4):273-285.
doi pubmed - Bateman BT, Schumacher HC, Wang S, Shaefi S, Berman MF. Perioperative acute ischemic stroke in noncardiac and nonvascular surgery: incidence, risk factors, and outcomes. Anesthesiology. 2009;110(2):231-238.
doi pubmed - McKhann GM, Grega MA, Borowicz LM, Jr., Baumgartner WA, Selnes OA. Stroke and encephalopathy after cardiac surgery: an update. Stroke. 2006;37(2):562-571.
doi pubmed - Bucerius J, Gummert JF, Borger MA, Walther T, Doll N, Onnasch JF, Metz S, et al. Stroke after cardiac surgery: a risk factor analysis of 16,184 consecutive adult patients. Ann Thorac Surg. 2003;75(2):472-478.
doi - Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.
doi pubmed - Biteker M, Kayatas K, Turkmen FM, Misirli CH. Impact of perioperative acute ischemic stroke on the outcomes of noncardiac and nonvascular surgery: a single centre prospective study. Can J Surg. 2014;57(3):E55-61.
doi pubmed - Dhamoon MS, Longstreth WT, Jr., Bartz TM, Kaplan RC, Elkind MSV. Disability trajectories before and after stroke and myocardial infarction: the cardiovascular health study. JAMA Neurol. 2017;74(12):1439-1445.
doi pubmed - Pearse RM, Moreno RP, Bauer P, Pelosi P, Metnitz P, Spies C, Vallet B, et al. Mortality after surgery in Europe: a 7 day cohort study. Lancet. 2012;380(9847):1059-1065.
doi - Deb S, Wijeysundera HC, Ko DT, Tsubota H, Hill S, Fremes SE. Coronary artery bypass graft surgery vs percutaneous interventions in coronary revascularization: a systematic review. JAMA. 2013;310(19):2086-2095.
doi pubmed - Jha AK, Li Z, Orav EJ, Epstein AM. Care in U.S. hospitals - the Hospital Quality Alliance program. N Engl J Med. 2005;353(3):265-274.
doi pubmed - Niessen LW, Barendregt JJ, Bonneux L, Koudstaal PJ. Stroke trends in an aging population. The Technology Assessment Methods Project Team. Stroke. 1993;24(7):931-939.
doi - Selim M. Perioperative stroke. N Engl J Med. 2007 Feb 15;356(7):706-13.
doi pubmed - Etzioni DA, Liu JH, Maggard MA, Ko CY. The aging population and its impact on the surgery workforce. Ann Surg. 2003;238(2):170-177.
doi pubmed - Lichtman JH, Leifheit-Limson EC, Jones SB, Watanabe E, Bernheim SM, Phipps MS, Bhat KR, et al. Predictors of hospital readmission after stroke: a systematic review. Stroke. 2010;41(11):2525-2533.
doi pubmed - Thirumala PD, Mohanraj SK, Habeych M, Wichman K, Chang YF, Gardner P, Snyderman C, et al. Value of free-run electromyographic monitoring of extraocular cranial nerves during expanded endonasal surgery (EES) of the skull base. J Neurol Surg Rep. 2013;74(1):43-50.
doi pubmed - Devereaux PJ, Sessler DI. Cardiac complications in patients undergoing major noncardiac surgery. N Engl J Med. 2015;373(23):2258-2269.
doi pubmed - Nogueira RG, Jadhav AP, Haussen DC, Bonafe A, Budzik RF, Bhuva P, Yavagal DR, et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med. 2018;378(1):11-21.
doi pubmed - Brott TG, Hobson RW, Howard G, et al. Stenting versus Endarterectomy for Treatment of Carotid-Artery Stenosis. N Engl J Med. 2010;363(1):11-23.
doi - Derdeyn CP, Chimowitz MI, Lynn MJ, Fiorella D, Turan TN, Janis LS, Montgomery J, et al. Aggressive medical treatment with or without stenting in high-risk patients with intracranial artery stenosis (SAMMPRIS): the final results of a randomised trial. Lancet. 2014;383(9914):333-341.
doi - Andrews RM. Statewide Hospital Discharge Data: Collection, Use, Limitations, and Improvements. Health Serv Res. 2015;50(Suppl 1):1273-1299.
doi pubmed - Fingar KR, Stocks C, Weiss AJ, Steiner CA. Most Frequent Operating Room Procedures Performed in U.S. Hospitals, 2003-2012: Statistical Brief #186. In: Healthcare Cost and Utilization Project (HCUP) Statistical Briefs. Rockville (MD), 2006.
- Nearman H, Klick JC, Eisenberg P, Pesa N. Perioperative complications of cardiac surgery and postoperative care. Crit Care Clin. 2014;30(3):527-555.
doi pubmed - Smilowitz NR, Gupta N, Ramakrishna H, Guo Y, Berger JS, Bangalore S. Perioperative major adverse cardiovascular and cerebrovascular events associated with noncardiac surgery. JAMA Cardiol. 2017;2(2):181-187.
doi pubmed - Goldman L, Caldera DL, Nussbaum SR, Southwick FS, Krogstad D, Murray B, Burke DS, et al. Multifactorial index of cardiac risk in noncardiac surgical procedures. N Engl J Med. 1977;297(16):845-850.
doi pubmed - Szeder V, Torbey MT. Prevention and treatment of perioperative stroke. Neurologist. 2008;14(1):30-36.
doi pubmed - WHO. Safe Surgery Saves Lives. World Heal Organ [Internet]. 2008;5(3):21. Available from: http://content.wkhealth.com/linkback/openurl?sid=WKPTLP:landingpage&an=01437870-200807000-00007.
doi - Sun Y, Paulus D, Eyssen M, Maervoet J, Saka O. A systematic review and meta-analysis of acute stroke unit care: what's beyond the statistical significance? BMC Med Res Methodol. 2013;13:132.
doi pubmed - Arboix A, Cartanya A, Lowak M, Garcia-Eroles L, Parra O, Oliveres M, Massons J. Gender differences and woman-specific trends in acute stroke: results from a hospital-based registry (1986-2009). Clin Neurol Neurosurg. 2014;127:19-24.
doi pubmed - Arboix A, Alio J. Acute cardioembolic stroke: an update. Expert Rev Cardiovasc Ther. 2011;9(3):367-379.
doi pubmed - Karamchandani K, Khanna AK, Bose S, Fernando RJ, Walkey AJ. Atrial fibrillation: current evidence and management strategies during the perioperative period. Anesth Analg. 2020;130(1):2-13.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.
