| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website http://www.neurores.org |
Case Report
Volume 6, Number 5-6, December 2016, pages 111-113
Clinical Effect of Leuprolide Acetate, an Agonist of GnRH, on Sensitive and Motor Function in a Patient With Chronic Spinal Cord Injury
J. Luis Quintanara, c, Maria del Carmen Diaz-Galindoa, Denisse Calderon-Vallejob, Irma Hernandez-Jassoa
aLaboratory of Neurophysiology, Department of Physiology and Pharmacology, Universidad Autonoma de Aguascalientes, Aguascalientes, Mexico
bDepartment of Morphology, Centro de Ciencias Basicas, Universidad Autonoma de Aguascalientes, Aguascalientes, Mexico
cCorresponding Author: J. Luis Quintanar, Depto. de Fisiologia y Farmacologia, Centro de Ciencias Basicas, Universidad Autonoma de Aguascalientes, Av. Universidad 940 C.P. 20131, Col. Ciudad Universitaria, Aguascalientes, Mexico
Manuscript accepted for publication November 23, 2016
Short title: Leuprolide Acetate in Spinal Cord Injury
doi: https://doi.org/10.14740/jnr403w
| Abstract | ▴Top |
The spinal cord is the major conduct through which sensory and motor signals pass between brain and body. Spinal cord injury (SCI) results in a disruption of these pathways and dramatic functional losses. There are different experimental approaches to counteracting the effects of SCI, such as the use of neurotrophic factors. It has been demonstrated that GnRH and its agonist leuprolide acetate (LA) have neurotrophic properties in SCI experimental model animals. Here, we report our findings from a patient suffering chronic SCI, who had undergone treatment with LA therapy. Treatment consisted of intramuscular injection of LA each month during 12 months. An important improvement in sensitive and motor functions was seen in the patient after treatment with LA. Further research is necessary to elucidate the mechanism of action of LA in SCI patients.
Keywords: Sensitive and motor functions; GnRH; Leuprolide acetate; Spinal cord injury; Neurotrophic factor; ASIA score
| Introduction | ▴Top |
Spinal cord injury (SCI) causes different and long-term neurological disorders and even morbidity in humans. The main types of neurological disorders after SCI include motor and sensory deficits, bladder, bowel and sexual dysfunction, chronic pain, and autonomic dysreflexia. The first approach in SCI is through surgical methods in conjunction with anti-inflammatory treatment as methylprednisolone [1]. There are different experimental approaches to counteracting the effects of SCI, such as the use of neurotrophic factors, Nogo neutralizing antibodies and N-metyl-D-aspartate receptor modulators [2, 3].
Recently, it has been demonstrated that GnRH and its agonist leuprolide acetate (LA) have neurotrophic properties. In vitro, GnRH incubation induced changes in outgrowth, number and length of neurites in rat spinal cord neurons [4]. In vivo, GnRH treatment improved locomotor activity and bladder function, and increased the expression of neurofilaments in spinal cords in rats with SCI [5]. Likewise, LA administration partially improves locomotor activity, gait, micturition reflex, and spinal cord morphology in a rat model of SCI [6]. LA is less susceptible to proteolysis than GnRH and has a greater binding affinity to receptors than the natural hormone, increasing its biological activity [7]. Currently, LA is used in patients for diverse clinical applications, including the treatment of prostate cancer, endometriosis, uterine fibroids, central precocious puberty and in vitro fertilization techniques [8]. LA administered via intramuscular is a non-invasive treatment and it is able to cross the blood-spinal cord barrier. It is safe for use and with few side effects, meaning a potential alternative for SCI treatment. Here, we report our findings from a patient suffering chronic SCI, who had undergone treatment with LA therapy.
| Case Report | ▴Top |
A 64-year-old male was admitted at rehabilitation clinic of the Autonomous University of Aguascalientes with complaints of being unable to walk, using wheelchair and with spasticity of lower limbs muscles since 3 years ago.
The subject was invited to voluntarily participate in the study. Before the initial assessment, he signed an informed consent, and all his questions or doubts about the study were clarified. This research was approved by the local Bioethical Committee of the Universidad Autonoma de Aguascalientes. All procedures used here were non-invasive and posed no hazard to the patient’s health.
The patient was diagnosed with incomplete lesion in T-9 with paralysis in lower limbs type C according to American Spinal American Association (ASIA) posterior to a vertebral column surgery. Physical examination revealed tightness in lower limbs and his lower limbs muscles were weak. He experienced difficulty in standing up without support. A detailed examination of the patient was done before and after the treatment. This treatment consisted of intramuscular injection of LA (3.75 mg) each month during 12 months. The patient has received rehabilitation from his lesion (3 years ago) and it was maintained during treatment with LA.
Before to LA administration, normal bilateral sensitivity was to T-9 and atypical sensitivity from T-10 to S-5 (Table 1). Changes in the level of sensitivity after treatment were observed (Table 1).
 Click to view | Table 1. Bilateral and Atypical Sensitivity of Patient Treated With LA (3.75 mg) Each Month During 12 Months |
Regarding the analysis of motor activity in hip flexors, knee extensors, ankle dorsiflexors, long toe externsors and ankle plantar flexors, results show a significant improvement (approximately 30%) at the end of 12 months of treatment with LA (Fig. 1). After treatment, spasticity was decreased assessed in heel reflex. At first of treatment, this reflex was absent, but at the final evaluation, it was presented with a hyporeflexia.
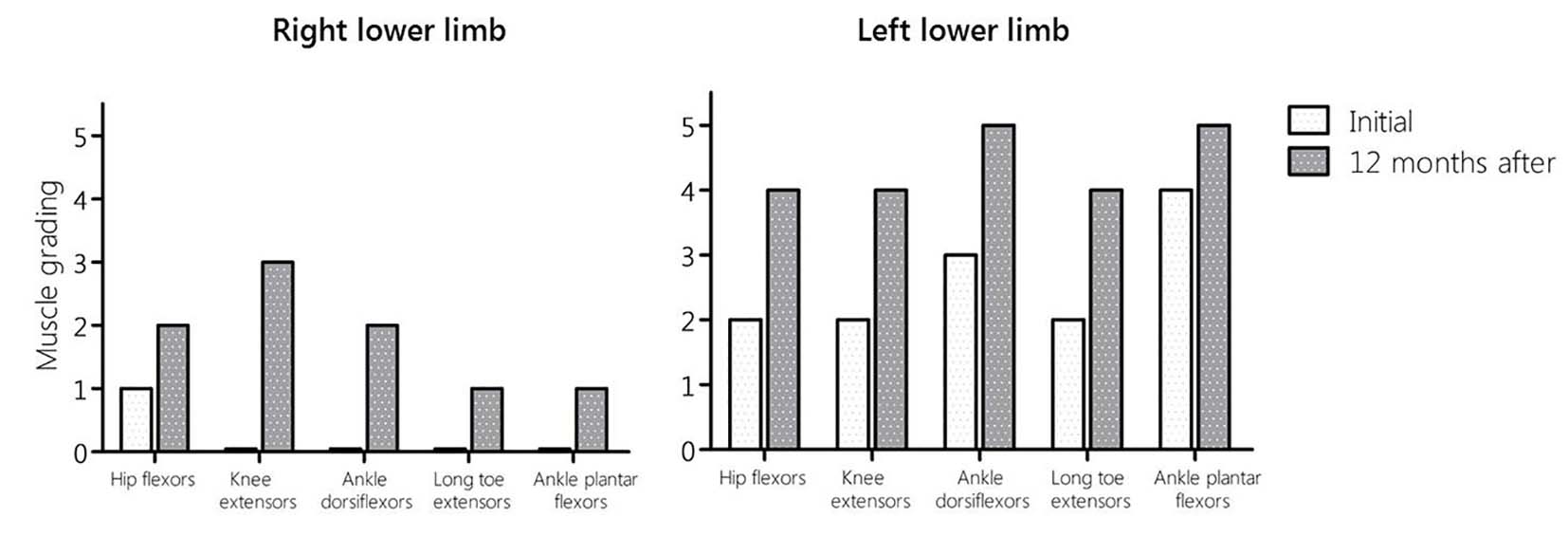 Click for large image | Figure 1. ASIA score results of motor activity in lower limbs before and after of 12 months of treatment with LA. |
In spinal cord independence measure, initially the patient was traveling in conventional wheelchair and can keep standing for seconds. However, 12 months after treatment with LA, he could walk slowly until 28 steps with the help of instruments and supervision.
| Discussion | ▴Top |
There are few reports on the administration of neurotrophic factors in patients with acute and chronic traumatic SCI. It has been reported that in patient with chronic SCI, there were significant improvements in ASIA motor and sensory scale scores and functional independence measure at 24 months after treatment with acidic fibroblast growth factor (aFGF) [9]. A difficulty of the procedure is that surgery is required for the administration of aFGF.
Similar results were found in patients with traumatic SCI who received treatment with thyrotropin-releasing hormone (TRH). In these patients, an intravenous dose of TRH was administered 12 h after SCI and sensory and motor functions were evaluated 4 months after treatment. The results of this study indicated a significantly higher sensory and motor recovery compared to placebo. In this case, TRH was injected acutely [10].
The present study provides a novel non-invasive treatment with LA, a GnRH agonist, to a patient with chronic SCI. Sensitive and motor functions were partially recovered with 12 months of treatment.
In SCIs, drug therapy for neurological recovery is a complex multifactorial process. Thus, according to the experimental models on neuroregeneration, LA administration could improve the conditions in patients with SCI through different mechanisms. It is possible that LA can induce: 1) an increase in the synthesis of protein basic of myelin, likewise a decrease in the factor NF-kB activation and mRNA expression levels of proinflammatory cytokines IL-1β, IL-17A and TNF-α as occurs in spinal cord of rats with experimental autoimmune encephalomyelitis [11, 12]; and 2) improvement in spinal cord morphology, locomotor behavior, gait, micturition reflex and reduction of microglial immunological reaction in spinal cord as it has been observed in a rat model of SCI [6].
LA injections were well tolerated. Furthermore, the administration of this agonist does not require a surgical procedure as in the case of aFGF, which is a great advantage for its use.
Conclusion
A significant sensitive and motor functions improvement was seen in a chronic SCI patient after LA therapy. The results must be interpreted with considerable caution because only a patient with chronic SCI was treated with LA, but together with several experimental animal studies, they support the need for a large multicenter clinical trial of LA.
Acknowledgments
The authors acknowledge Dr. Andres Quintanar Stephano for his medical assistance, Lic. Luis Ernesto Munoz for his support in the Physical Rehabilitation Clinic of the Universidad Autonoma de Aguascalientes and Dr. Ernesto Ruiz Esparza Padilla and Ing. Victor Masaaki Ono Yoshikawa for their support in the Medical Center Medica Norte.
Conflicts of Interest
The authors do not have any conflicts of interest.
| References | ▴Top |
- Varma AK, Das A, Wallace Gt, Barry J, Vertegel AA, Ray SK, Banik NL. Spinal cord injury: a review of current therapy, future treatments, and basic science frontiers. Neurochem Res. 2013;38(5):895-905.
doi pubmed - Thuret S, Moon LD, Gage FH. Therapeutic interventions after spinal cord injury. Nat Rev Neurosci. 2006;7(8):628-643.
doi pubmed - Harvey AR, Lovett SJ, Majda BT, Yoon JH, Wheeler LP, Hodgetts SI. Neurotrophic factors for spinal cord repair: Which, where, how and when to apply, and for what period of time? Brain Res. 2015;1619:36-71.
doi pubmed - Quintanar JL, Calderon-Vallejo D, Hernandez-Jasso I. Effects of GnRH on Neurite Outgrowth, Neurofilament and Spinophilin Proteins Expression in Cultured Spinal Cord Neurons of Rat Embryos. Neurochem Res. 2016;41(10):2693-2698.
doi pubmed - Calderon-Vallejo D, Quintanar JL. Gonadotropin-releasing hormone treatment improves locomotor activity, urinary function and neurofilament protein expression after spinal cord injury in ovariectomized rats. Neurosci Lett. 2012;515(2):187-190.
doi pubmed - Diaz Galindo C, Gomez-Gonzalez B, Salinas E, Calderon-Vallejo D, Hernandez-Jasso I, Bautista E, Quintanar JL. Leuprolide acetate induces structural and functional recovery of injured spinal cord in rats. Neural Regen Res. 2015;10(11):1819-1824.
doi pubmed - Periti P, Mazzei T, Mini E. Clinical pharmacokinetics of depot leuprorelin. Clin Pharmacokinet. 2002;41(7):485-504.
doi pubmed - Wilson AC, Meethal SV, Bowen RL, Atwood CS. Leuprolide acetate: a drug of diverse clinical applications. Expert Opin Investig Drugs. 2007;16(11):1851-1863.
doi pubmed - Wu JC, Huang WC, Tsai YA, Chen YC, Cheng H. Nerve repair using acidic fibroblast growth factor in human cervical spinal cord injury: a preliminary Phase I clinical study. J Neurosurg Spine. 2008;8(3):208-214.
doi pubmed - Pitts LH, Ross A, Chase GA, Faden AI. Treatment with thyrotropin-releasing hormone (TRH) in patients with traumatic spinal cord injuries. J Neurotrauma. 1995;12(3):235-243.
doi pubmed - Guzman-Soto I, Salinas E, Hernandez-Jasso I, Quintanar JL. Leuprolide acetate, a GnRH agonist, improves experimental autoimmune encephalomyelitis: a possible therapy for multiple sclerosis. Neurochem Res. 2012;37(10):2190-2197.
doi pubmed - Guzman-Soto I, Salinas E, Quintanar JL. Leuprolide Acetate Inhibits Spinal Cord Inflammatory Response in Experimental Autoimmune Encephalomyelitis by Suppressing NF-kappaB Activation. Neuroimmunomodulation. 2016;23(1):33-40.
doi pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.
