| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website http://www.neurores.org |
Case Report
Volume 10, Number 2, April 2020, pages 44-47
Severe Cerebellar Syndrome Linked With Durvalumab
Connie G. Tanga, c, Goran Rakocevicb, c
aDepartment of Neurology, Thomas Jefferson University, Philadelphia, PA, USA
bDepartment of Neurology, University of Virginia Health System, Charlottesville, VA, USA
cCorresponding Author: Connie G. Tang, 901 Walnut Street, 4th Floor, Philadelphia, PA 19107, USA; Goran Rakocevic, Department of Neurology, University of Virginia Health System, Charlottesville, VA 22903, USA
Manuscript submitted February 4, 2020, accepted February 14, 2020
Short title: Cerebellar Syndrome Linked With Durvalumab
doi: https://doi.org/10.14740/jnr571
| Abstract | ▴Top |
Durvalumab is a monoclonal antibody that blocks the interaction of programmed cell death ligand 1 (PD-L1) with programmed cell death 1 (PD-1) molecules. It has shown significant survival benefits in various cancers as an immune checkpoint inhibitor. With increased usage of this therapy, there are several cases with a predilection for neurological immune-related adverse effects from checkpoint inhibitor neurotoxicity. We identified an index patient with lung cancer treated with durvalumab, who developed disabling ocular and bulbar symptoms and severe truncal ataxia, and found symptomatic benefit with plasmapheresis (PLEX). A 57-year-old African American woman presented with 3 months of oscillopsia, dizziness, scanning speech, and inability to stand and walk due to severe truncal ataxia. She was found to have stage IIIA lung adenocarcinoma. Brain magnetic resonance imaging (MRI) demonstrated abnormal T2-fluid-attenuated inversion recovery sequence (T2/FLAIR) signal in bilateral cerebellar hemispheres with lack of enhancement suggestive of paraneoplastic cerebellar involvement. No distinct paraneoplastic antibody was identified. She received combined pulse-dose steroids and intravenous immunoglobulin (IVIg) with no improvement of cerebellar syndrome. A month later, she started inpatient chemotherapy and concurrent radiation therapy with transient cancer regression. Because of subsequent metastatic spread, durvalumab was initiated. She completed four doses that were complicated by worsening cerebellar symptoms and autoimmune colitis. During durvalumab holiday, she received two courses of five PLEX treatments, 2 months apart, along with vigorous physical and speech therapy. Her neurologic symptoms and functional status improved considerably and continued to improve after treatment. At present time, the patient is largely independent for activities of daily living (ADLs) and uses a walker. Repeated brain MRI revealed resolution of previously seen abnormalities. Checkpoint inhibitors may worsen concomitant paraneoplastic neurologic syndromes that develop in association with malignancies potentially responsive to PD-L1 and PD-1 inhibitors. Meticulous coordination and timing of life-saving immune therapies for cancer with effective immune treatments for an underlying or associated neurological syndrome are essential for best outcomes.
Keywords: All clinical neurology; MRI; All medical/systemic disease; Gait disorders/ataxia; Paraneoplastic syndrome
| Introduction | ▴Top |
Durvalumab is a monoclonal antibody that blocks the interaction of programmed cell death ligand 1 (PD-L1) with programmed cell death 1 (PD-1) molecules. It has shown significant survival benefits in various cancers as an immune checkpoint inhibitor (ICI). With increased usage of this consolidation therapy for cancers, there have been several reported cases with a predilection for neurological immune-related adverse effects (irAEs) from checkpoint inhibitor neurotoxicity. We identified an index patient with lung cancer treated with durvalumab, who developed disabling ocular and bulbar symptoms and severe truncal ataxia, and found symptomatic benefit with plasmapheresis (PLEX). We discuss our patient findings as well as implications on therapy with PLEX and timing of administration.
| Case Report | ▴Top |
A 57-year-old African American woman with a history of 35 pack-years of smoking and emphysema subacutely developed dizziness, change of speech and gait imbalance. After several unprotected falls, the patient was evaluated in the emergency department. Exam revealed a scanning speech, persistent multidirectional nystagmus with rotatory component, and bilateral dysmetria with severe truncal ataxia. She was also noted to have 15 - 20 pounds of unintentional weight loss over a period of several months. Brain magnetic resonance imaging (MRI) demonstrated bilateral cerebellar T2 hyperintensities without enhancement (Fig. 1a, b) consistent with a paraneoplastic cerebellar syndrome in the setting of newly diagnosed stage III non-small cell lung cancer (NSCLC). Lumbar puncture was performed revealing 45 white blood cells (WBCs) with lymphocytic predominance and mildly elevated protein level at 69 mg/dL. Flow cytometry did not reveal atypical cells, and no paraneoplastic antibody was identified in both serum and cerebrospinal fluid (CSF). Patient received intravenous (IV) steroids (1 g daily for 5 days) followed by intravenous immunoglobulin (IVIg) (2 g/kg) with short-lived improvement of cerebellar symptoms. After a full course of conventional chemotherapy for NSCLC, severe cerebellar symptoms persisted resulting in patient’s inability to stand and walk. She then underwent five plasma exchanges (PEs) with gradual improvement of dysarthria and truncal ataxia in ensuing 3 months. Repeated brain MRI demonstrated resolution of cerebellar hyperintensities (Fig. 1c, d). Surveillance imaging showed a persisting cancer with diminished mediastinal and hilar adenopathy.
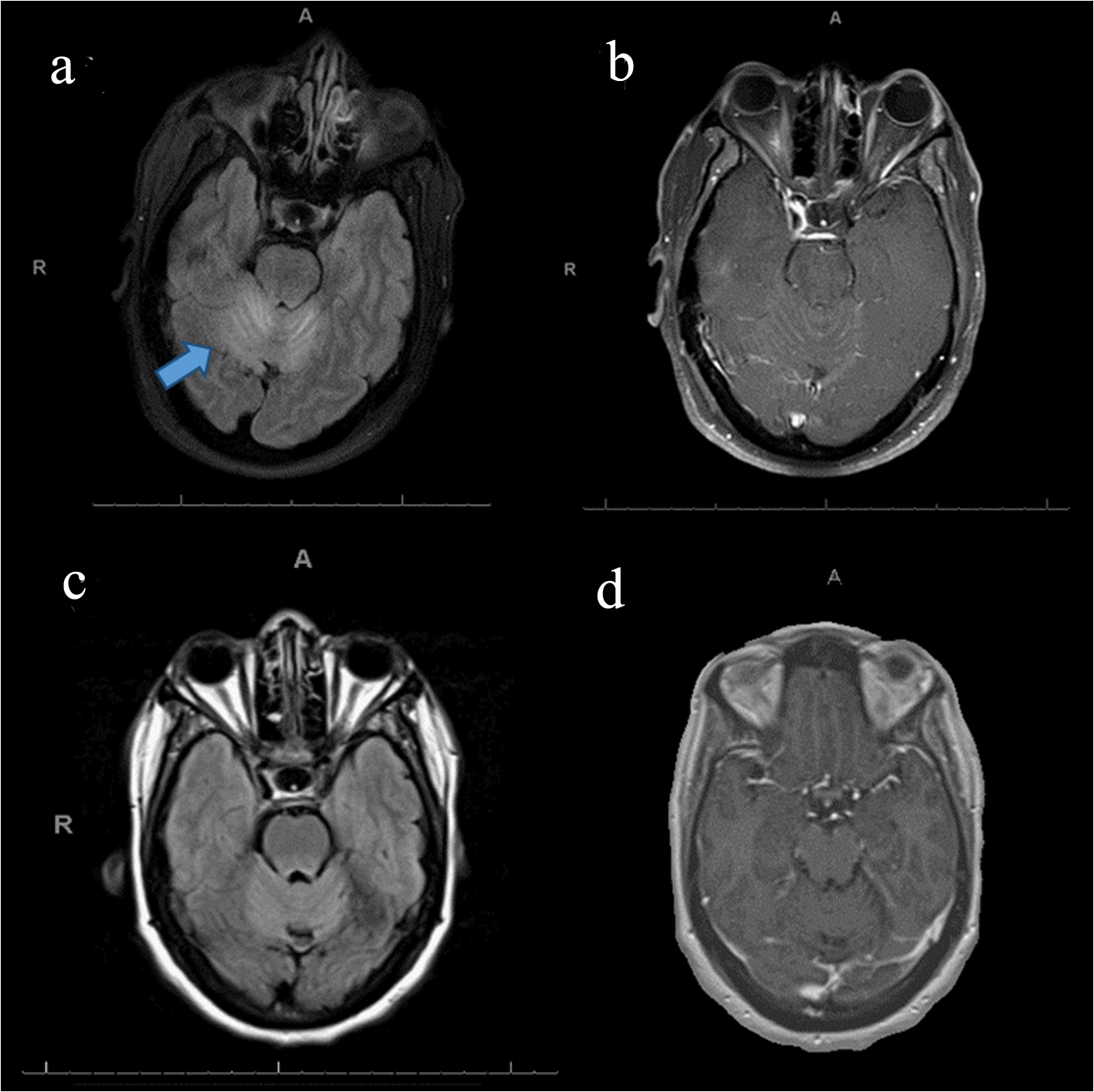 Click for large image | Figure 1. Brain MRI before and after first plasmapheresis. (a) Abnormal T2/FLAIR signal in bilateral cerebellar hemispheres, (b) post-contrast study performed at the same time as (a) showing lack of contrast enhancement, (c) resolution of previously seen signal abnormality in the cerebellum, and (d) post-contrast study performed at the same time as (c) with no change. MRI: magnetic resonance imaging; T2/FLAIR: T2-fluid-attenuated inversion recovery sequence. |
Patient was eventually recommended durvalumab, a PD-L1 checkpoint inhibitor, as cancer consolidation therapy. Subsequently, after three cycles, she developed autoimmune colitis and was started on prednisone 20 mg daily. Two months later, she had marked worsening of gait ataxia, oscillopsia and dysarthria. Repeated contrast MRI of brain did not reveal enhancement, and both neuroimaging and cancer screening were negative for a metastatic process. Checkpoint inhibitor was discontinued for 3 weeks in the context of severe neurological and gastrointestinal (GI) complications. This allowed for another trial of PLEX followed by inpatient rehabilitation program, which was beneficial in improving her speech, dysmetria as well as gait ataxia and stamina. This allowed for resuming durvalumab 4 weeks later. However, after 3 months of additional ICI therapy, patient’s cerebellar symptoms worsened again necessitating durvalumab discontinuation. Three weeks after discontinuation, a third series of PLEX was administered with improvement of her speech and gait to where she was able to walk short distances unassisted for next 4 - 6 months. Two years after the initiation of ICI, patient developed durvalumab-associated dermatitis, brittle diabetes, and incessant colitis. Thus, durvalumab was stopped indefinitely.
| Discussion | ▴Top |
In the last several years, the cancer field has embraced ICIs as means to either augment the immune system or appropriate the body’s own defense system against cancer cells. ICIs have become a promising class of anticancer therapy by acting as a “switch” to turn on heightened surveillance for foreign cells. Specifically, PD-1 is a checkpoint protein on T cells that helps prevent T cells from attacking other cells in the body when it attaches to PD-L1 [1]. Some cancer cells have large amounts of PD-L1, which helps them evade immune attack [1]. ICIs target either PD-1 or PD-L1 proteins, thereby switching on an immune response to these cells. In this case discussion, durvalumab is a PD-L1 inhibitor used as adjunctive therapy in NSCLC [2].
However, there is a growing body of evidence for severe irAEs accompanying ICI therapy. Up to 70% of patients undergoing treatment with PD-1/PD-L1 inhibitors are reported to have experienced some irAEs [3]. These are believed to be due to a nonspecific immune checkpoint inhibition disrupting the regulatory homeostasis against autoimmunity, leading to various local and systemic autoimmune responses. Neurological complications of PD-1 inhibitors have been estimated with a frequency of 4.2% [4]. Several neurologic adverse events have been described, including encephalitis, myelopathy, aseptic meningitis, meningoradiculitis, Guillain-Barre-like syndrome, peripheral neuropathy, myopathy, as well as myasthenic syndrome [5]. In a large cohort study by Kao et al, they examined 373 patients undergoing PD-1 therapy for a solid tumor and 10 patients were identified to have a neurological complication (2.9%) [4]. Of those 10, one was identified to have a cerebellar ataxia and dysarthria, who improved after cessation of therapy [4]. Most irAEs occur within 3 - 6 months of starting a PD-1/PD-L1 inhibitor [5]. Although, in some instances irAEs may occur after the first treatment, they can be delayed, or happen in variable timeline after cessation of immunotherapy [6].
Recognition and appropriate treatment of neurological symptoms in patients undergoing ICI therapy are crucial as this may lead to severe neurologic disability and even death. There is limited information regarding outcomes of irAEs in various cancers. Therefore, consideration must balance the risk of decreasing efficacy of immune checkpoint therapy if treatment is withheld, and the benefit of immunomodulating/suppressive therapy for neurological symptoms that may interfere with cancer treatments and/or potentially alter the patient’s immune status unfavorably leading to additional complications. Early recognition and timely treatment of neurological syndrome though may offer a chance at neurological deficit reversal and improved overall outcome [7].
In the case of ICI-associated neurological complications, therapeutic strategy most commonly involves cessation of the ICI, with the initiation of IV or oral steroids combined with supportive therapies. This approach provided a complete resolution in a majority of cases; however, many patients developed irreversible adverse events including deaths reported in a few cases [8]. In steroid-refractory cases, immunomodulatory treatments with PLEX or IVIg may be effective [9]. While the frequency and duration of such immunomodulatory therapies have not been well established, with the increasing use of ICIs it is observed that repeated PEs or IVIg may be necessary for exacerbations of neurologic symptoms. Although our patient had developed paraneoplastic neurological syndrome prior to the initiation of ICIs, her cerebellar symptoms and gait invariably worsened within days of each durvalumab course. We pursued carefully timed PLEX as the patient’s symptoms had been refractory to IVIg therapy previously.
Timing and frequency of PEs for worsening neurological syndrome related to ICIs have also been an unsettled issue. The sooner immunomodulating therapy for neurological complications begins, the better clinical response is expected. The concern remains on whether neuro-immunomodulating therapies such as PE and IVIg will diminish the efficacy of the checkpoint inhibitor for the underlying cancer. In general, this question is still approached empirically. Since the half-life of ICIs is estimated between 2 and 4 weeks and in order to strike the balance between efficacy and safety, PEs are not recommended to follow ICIs immediately. We chose a time period of approximately 3 weeks after the last durvalumab infusion to begin PLEX as to not compromise the ICI’s efficacy. Nevertheless, if the patient’s neurological status is unstable and deteriorating, appropriate immunomodulating treatment needs to be considered and provided sooner. Ideally, the individualized treatment plan is devised and adjusted among oncologist, neurologist and other treating teams.
Continual clinical vigilance of the interdisciplinary teams caring for patient undergoing ICI therapy is of paramount importance. Early recognition and effective treatments of ICI-associated neurological and other complications provide the best potential for reducing morbidity and deaths, and for improving patients’ functional status and quality of life. Established clinical guidelines from growing case series and prospective studies of adverse events are needed to aid in the treatment choices of ICIs and the management of related irAEs.
Acknowledgments
The authors thank the Jefferson Department of Oncology in coordination of care of this patient and many others.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Subject has given informed consent.
Author Contributions
CGT drafted manuscript for intellectual content; GR drafted and revised the manuscript.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Dalakas MC. Neurological complications of immune checkpoint inhibitors: what happens when you 'take the brakes off' the immune system. Ther Adv Neurol Disord. 2018;11:1756286418799864.
doi pubmed - Fellner A, Makranz C, Lotem M, Bokstein F, Taliansky A, Rosenberg S, Blumenthal DT, et al. Neurologic complications of immune checkpoint inhibitors. J Neurooncol. 2018;137(3):601-609.
doi pubmed - Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, Powderly JD, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366(26):2443-2454.
doi pubmed - Kao JC, Liao B, Markovic SN, Klein CJ, Naddaf E, Staff NP, Liewluck T, et al. Neurological complications associated with anti-programmed death 1 (PD-1) antibodies. JAMA Neurol. 2017;74(10):1216-1222.
doi pubmed - Astaras C, de Micheli R, Moura B, Hundsberger T, Hottinger AF. Neurological adverse events associated with immune checkpoint inhibitors: diagnosis and management. Curr Neurol Neurosci Rep. 2018;18(1):3.
doi pubmed - Weber JS, Hodi FS, Wolchok JD, Topalian SL, Schadendorf D, Larkin J, Sznol M, et al. Safety profile of nivolumab monotherapy: a pooled analysis of patients with advanced melanoma. J Clin Oncol. 2017;35(7):785-792.
doi pubmed - Khoja L, Day D, Wei-Wu Chen T, Siu LL, Hansen AR. Tumour- and class-specific patterns of immune-related adverse events of immune checkpoint inhibitors: a systematic review. Ann Oncol. 2017;28(10):2377-2385.
doi pubmed - Bajwa R, Cheema A, Khan T, Amirpour A, Paul A, Chaughtai S, Patel S, et al. Adverse effects of immune checkpoint inhibitors (Programmed death-1 inhibitors and cytotoxic T-lymphocyte-associated protein-4 inhibitors): results of a retrospective study. J Clin Med Res. 2019;11(4):225-236.
doi pubmed - Hottinger AF. Neurologic complications of immune checkpoint inhibitors. Curr Opin Neurol. 2016;29(6):806-812.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.
