| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website http://www.neurores.org |
Original Article
Volume 4, Number 1, February 2014, pages 22-30
Learning Disabilities in Different Types of Attention Deficit Hyperactivity Disorders and its Relation to Cortical and Brainstem Function
Mohamed A. Ahmeda, c, Khaled A. El-Beha, Talal A. Mohammadb, Dina F. Mansourb, Mohammed Y. Ezz-Eldineb
aDepartment of Neuropsychiatry, Faculty of Medicine, Assiut University Hospital, Assiut, Egypt
bDepartment of Neuropsychiatry, Faculty of Medicine, Qena University Hospital, Qena, Egypt
cCorresponding author: Mohamed Abdel-Rahman Ahmed, Department of Neurology, Assiut University Hospital, Assiut, Egypt
Manuscript accepted for publication February 26, 2014
Short title: Attention Deficit Hyperactivity Disorders
doi: https://doi.org/10.14740/jnr268w
| Abstract | ▴Top |
Background: Attention deficit hyperactivity disorder (ADHD) is comprised of the triad of inattention, hyperactivity and impulsivity. It was hypothesized that ADHD children and adults have abnormalities not only in several late-developing fronto-striatal networks but also in temporal-parietal and fronto-cerebellar neural networks which mediate the cognitive control functions that are impaired in this disorder. So the aim of this study is to determine the learning disabilities in different types of ADHD and its relation to brainstem and cortical function.
Patients and Methods: Forty children participated in the study. All children were subjected to a full neurological and psychiatric examination: Swanson, Nolan and Pelham Questionnaire 4th edition (SNAP-IV-1992), Myklebust learning disability scale, auditory brainstem response (ABR) and electroencephalography (EEG).
Results: Patients with ADHD showed significant lower level of learning disability score, and the inattentive type had the worst score. The mean of ABR had significant delay in wave III, IV, V, I-III and I-V interval, and the delay was significantly high in inattentive type. Fifteen (37.5%) children with ADHD had abnormal non-epileptiform activity and five (15%) had epileptiform activity. Inattentive type showed the highest abnormal activities. Learning disabilities showed non-significant negative correlation to score of inattention type, significant negative correlation to absolute latency of wave III, IV, V, interpeak latency between wave I-III and I-V interval and significant negative correlation to epileptiform activity in EEG.
Conclusion: Learning disabilities are strongly co-morbid with ADHD especially inattentive type and both may had brainstem and cortical processing abnormalities.
Keywords: Attention deficit hyperactivity disorders; Learning disabilities; ABR; EEG
| Introduction | ▴Top |
Attention deficit hyperactivity disorder (ADHD) is comprised of the triad of inattention, hyperactivity and impulsivity. It is one of the most common childhood-onset neurodevelopment disorders, with a prevalence of around 5% in children [1] and 3% in adults [2]. While the onset is usually before the age of 7 years, a majority (up to 65% of cases) has persistent, impairing symptoms into adulthood [3].
It was hypothesized that ADHD children and adults have abnormalities not only in several late-developing fronto-striatal networks but also in temporal-parietal and fronto-cerebellar neural networks which mediate the cognitive control functions that are impaired in this disorder [4, 5].
Learning disorders (LDs) affect about 2-10% of the school-age population. They are characterized by an academic functioning that is below the level that would be expected given their age, intelligent quotient and grade level in school, and interfere significantly with academic performances or daily life activities that require reading, writing or calculation skills [6]. Giraud and Ramus [7] described a putative mechanistic model that linked neuronal micro-architecture of the auditory cortex to specific alterations of phonological processing. They suggested that dyslexia could be related to a disconnection syndrome and associated with neuroanatomical alterations, involving both the white and the gray matter of a frontotemporo-parietal network, suggestive of dysfunction in cortical connectivity.
ADHD is associated with significant academic, behavioral and social impairment throughout the life span [8, 9].
Central auditory processing disorder (CAPD) has been a debate about its relation to ADHD. Although the co-morbidity of CAPD with ADHD has been well documented [10], some researchers argued that CAPD and ADHD may be overlapping but independent disorders [11]. There is a similarity between ADHD and CAPD in symptomatology as well as in psychoeducational and behavioral sequelea [12]. Tillery et al [13] concluded that a diagnosis of ADHD places the child at risk (50-80%) for CAPD.
Auditory evoked potentials are considered biological markers that provide information about neural timing with fractions of millisecond precision [14]. Auditory brainstem response (ABR) provides information about the functional integrity of brainstem nuclei along the ascending auditory pathway up to midbrain inferior colliculus [15].
The relationship between epilepsy and ADHD is complex and not well understood [16, 17]. There is some evidence that children with ADHD have a higher rate of interictal epileptiform abnormalities on electroencephalography (EEG) compared with those without ADHD according to several studies [18, 19].
So the aim of this study is to determine the learning disabilities in different types of ADHD and its relation to brainstem and cortical function.
| Patient and Methods | ▴Top |
Forty children (25 males and 15 females) participated in the study; they were collected from the outpatient clinic of psychiatry between December 2012 and May 2013. Age of children ranged between 6 and 12 years old, fulfilling the DSM-IV criteria for ADHD and never received treatment for ADHD. Children with intelligent question more than or equals 90 were included. Children with other co-morbid general medical or neurological illness were excluded. All children were found to have negative history for maternal exposure to smoking, drugs, toxins or alcohol during pregnancy. They all had normal natal, postnatal and developmental history.
Twenty normal children volunteers participated in the study from general population. The mean age for patients was 7.68 ± 1.64 years; 25 (62.5%) of them were males and 15 (37.5%) were females. The control group consisted of nine (45%) males and 11 (55%) females and the mean age was 8.05 ± 1.73 years. There was no statistically significant difference between the two groups regarding age and sex.
All caretakers of the children gave written consent to participate their children in the study after full explanation of the study procedures was provided.
Methods
All children were subjected to the followings.
A full neurological and psychiatric history was obtained from all children and full general and neurological examination was done.
Swanson, Nolan and Pelham Questionnaire 4th edition (SNAP-IV-1992) [20, 21] which is designed semi-structured interview was done. Subscale scores are calculated by summing the scores of the items and dividing by the number of the items. We used the parent edition. Cutoffs are as follows: 1.78 for ADHD-I, 1.44 for ADHD-HI and 1.67 for ADHD-C. This scale is very helpful in diagnosis as well as measuring severity of ADHD symptoms.
According to the DSM-IV, and Swanson, Nolan and Pelham Questionnaire 4th edition (SNAP-IV-1992) scale, children were categorized into three subtypes of the ADHD: predominantly inattentive (ADHD-I), predominantly hyperactive-impulsive (ADHD-HI) and combined type (ADHD-C).
Modified Myklebust learning disability scale was used to determine the learning affection in the diseased and control groups; it consists of 24 items and its cut point is 1.98. The scale was translated into Arabic language and standardized in Jordon which is an Arabic Country like Egypt [22].
Neurophysiologic studies: 1) Click ABR was carried out for patients and control group with alternating click of intensity 90 dB delivered by an earphone, with 10 pulses/s as repetition rate. We recorded the absolute latencies of wave I, II, III, IV and V and the interwave intervals of I-III, III-V and I-V on both sides. 2) Digitalized routine EEG was obtained from all children using eight-channel Nihon Kohden machine employing scalp electrodes placed according to the international 10-20 system with bipolar and referential montages. EEG interpretation was done by professor of neurology in two separate sessions. The interpretation was classified either to normal EEG, abnormal non-epileptiform EEG and epileptiform activity.
Statistical analysis
Statistical values were expressed as mean ± standard deviation (SD) using SPSS program version 17. These results were analyzed statistically using the independent Student’s t-test. Pearson correlation coefficient (r) was used to measure correlation between quantitative variables. Chi-square test and Spearman correlation coefficient were used to measure differences and correlations between qualitative variables respectively. Comparisons among more than two groups for differences in estimated means were conducted with analysis of variance (ANOVA) test.
| Results | ▴Top |
In the studied patients, 28 children (70%) had combined ADHD, inattentive type in nine children (22.5%) and hyperactive-impulsive type in three children (7.5%) with male to female ratio 15:13, 7:2 and 3:0 respectively.
Patients with ADHD showed significant lower level of learning disability score in patient in comparison to control group (P < 0.000) (Table 1). The inattentive type had the worst score in comparison to combined and hyperactive-impulsive type (P = 0.019) (Table 2, Fig. 1).
 Click to view | Table 1. Comparison of Learning Disability Scores in Patient and Control Groups |
 Click to view | Table 2. Comparison of Learning Disability Scores in ADHD Subtypes |
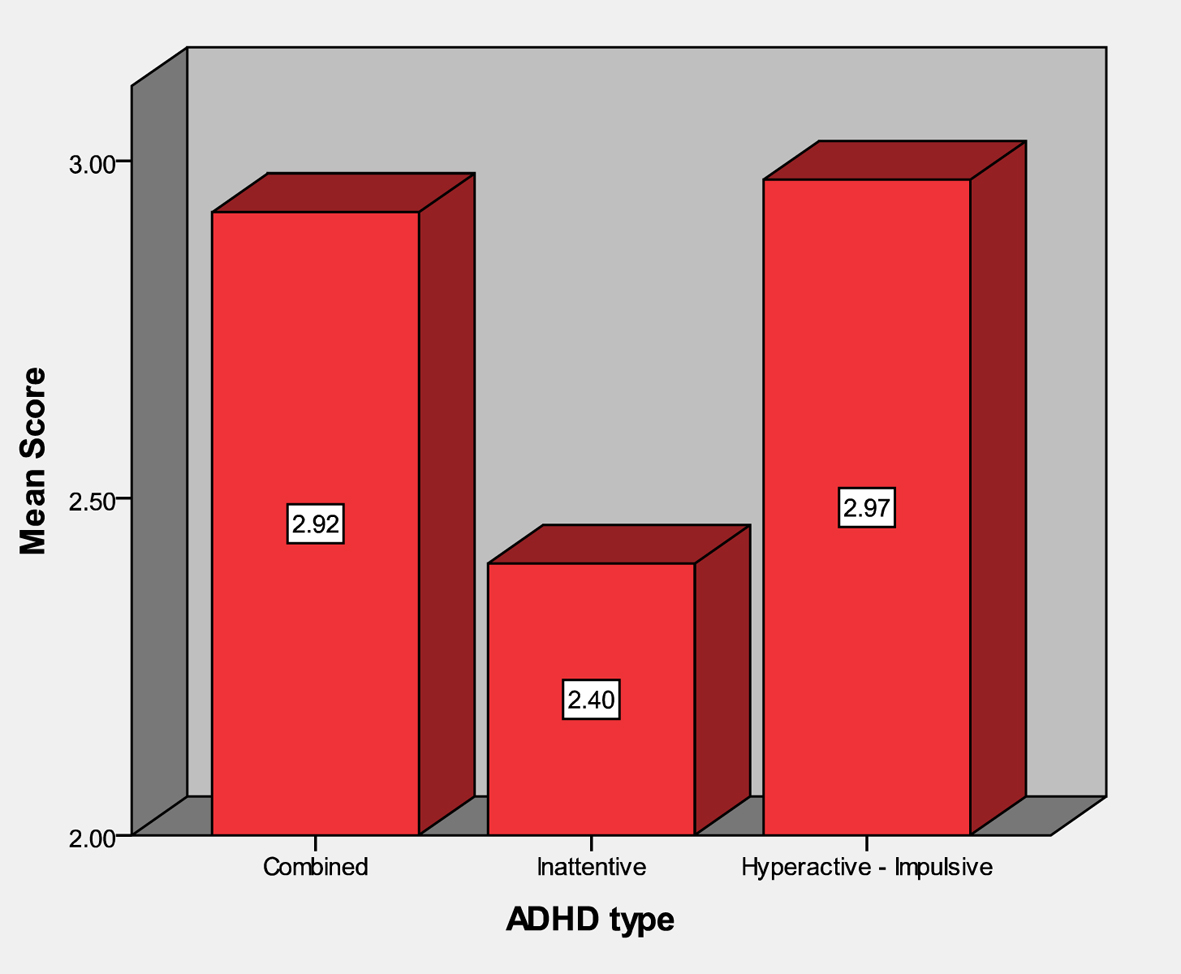 Click for large image | Figure 1. Comparison of learning disability scores in ADHD subtypes. |
As regard ABR in the studied patients, there was no significant difference between right and left side in absolute latency of ABR waves and intervals in patients and control groups. So we take the mean of both right and left side in patients and compared with the mean of both right and left side in control.
The mean of ABR waves and intervals of patients had significant delay in wave III (P = 0.025), wave IV (P = 0.018), wave V (P = 0.006), I-III interval and I-V interval (P = 0.001) and III-V (P = 0.035) in comparison to control (Table 3). The delay was significantly high in inattentive type in wave III (P = 0.000), wave V (P = 0.002), I-III interval (P = 0.001) and I-V interval (P = 0.002) in comparison to the combined and the hyperactive impulsive type (Table 4).
 Click to view | Table 3. Comparison Between the ABR Means of Both Ears in Case and Control Groups |
 Click to view | Table 4. One-Way ANOVA Test Comparing Means of ABR Waves and Intervals in ADHD Subtypes |
Nineteen (47.5%) of the studied children had normal EEG, 15 (37.5%) children with abnormal non-epileptiform activity and five (15%) children with epileptiform activity. Inattentive type showed two (22.2%) children with normal EEG, three (33.3%) children with abnormal non-epileptiform EEG and four (44.4%) children with epileptiform EEG activity. The combined type showed 16 (57.1%) children with normal EEG, 11 (39.3%) children with abnormal non-epileptiform EEG and one (3.6%) child with epileptiform EEG activity. Hyperactive type showed one (33.3%) child with normal EEG, one (33.3%) child with abnormal non-epileptiform EEG and one (33.3%) child with epileptiform EEG activity (Fig. 2).
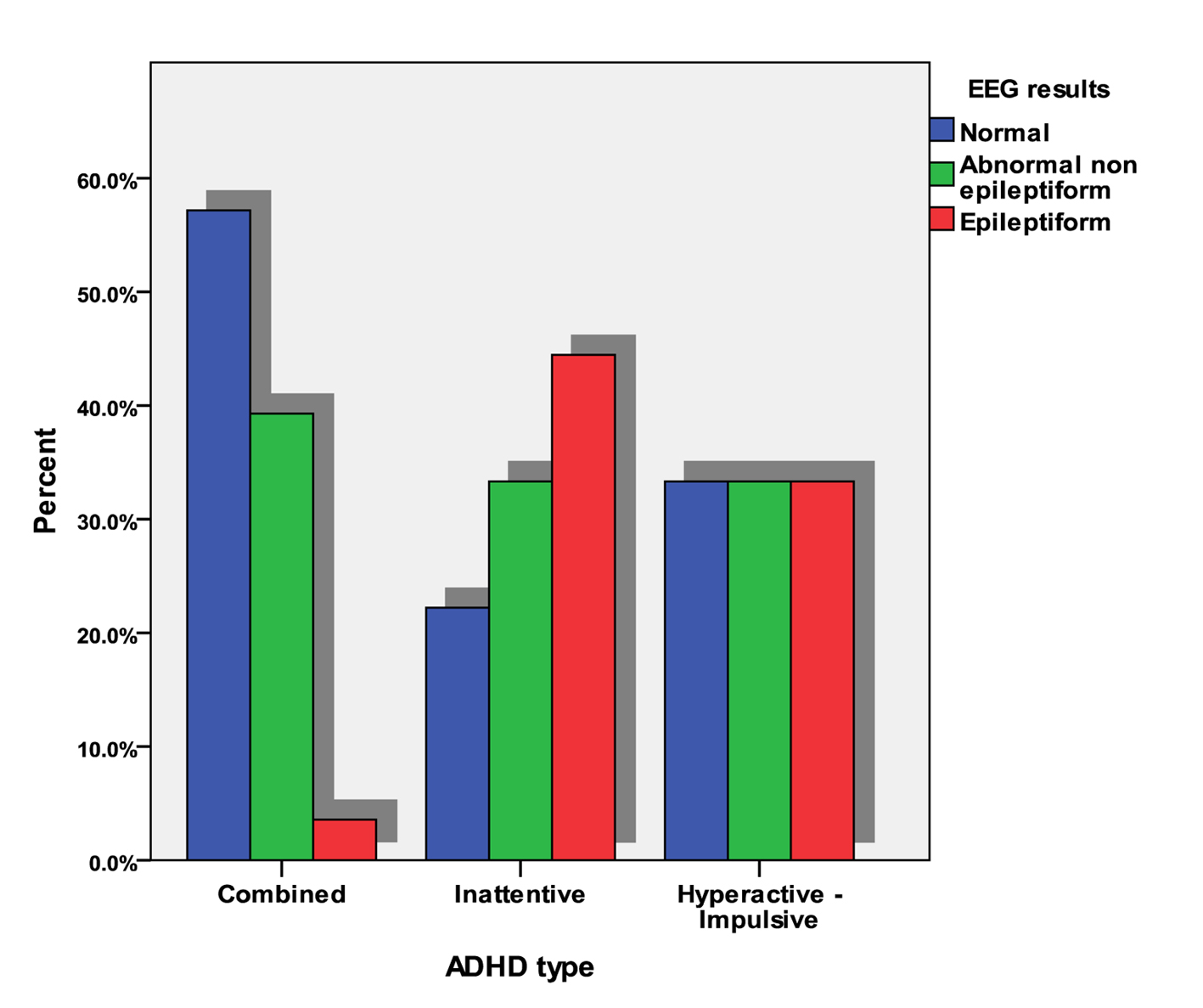 Click for large image | Figure 2. Distribution of normal and abnormal EEG within ADHD groups. |
Learning disabilities showed strong negative correlation to score of inattention type according to SNAP IV ADHD scale (r = -0.310; P = 0.051). Regarding neurophysiologic parameters, learning disabilities had significant negative correlation to absolute latency of wave III, IV, V, interpeak latency between wave I-III and I-V interval. Learning disabilities also had significant negative correlation to epileptiform activity in EEG (Table 5).
 Click to view | Table 5. Correlations of Learning Disability to Neurophysiology Parameters |
| Discussion | ▴Top |
Our study showed significant lower score in learning abilities in children with ADHD in comparison to control. Old and less replicated studies have suggested that reading disorder might be the primary deficit which causes secondary symptoms of ADHD [23-26]. Co-morbidity with ADHD is present from 10% to 50% of LDs children, while co-morbidity with dyslexia is present from 25 to 40% of ADHD patients [27-30]. Recent data have shown that there are common cognitive deficits between the ADHD and learning disability [31] according to a possible similar genetic etiology, as demonstrated by families’ studies in twins [32, 33]. Margari et al’s [6] results support these latter theories, as demonstrated by the higher frequency of ADHD in SLD patients as they reported that ADHD combined or isolated is present in 63 (33%), 55 males and eight females with LD. Motor coordination disorder was reported in a percentage from 10.3% to 26% of dyslexics [34, 35]. These data support the “cerebellar theory” of dyslexia [36] according to which, the cerebellum, that is responsible for motor control and automate over learned tasks (namely, reading), in LD may exert an insufficient motor control influencing articulation, phonological representation and ability to form appropriate connections between graphemes and phonemes. Carlson and Mann [37] mentioned that no consistent significant differences have been found between children with the combined and inattentive types of ADHD in cognitive and academic achievement tests, although both tend to have poorer performance than controls. In the present work, inattentive type had the lowest score in learning ability in comparison to other two types but no previous studies reported LDs in different types of ADHD.
In the present study, there was significant prolongation of absolute and interpeak latencies of waves (III, IV and V) of ABR in both sides with delay in transmission of impulses from the auditory nuclei in low brainstem (interval I-III) and rostral midbrain (interval III-V) and total brainstem transmission time (interval I-V) in ADHD patients compared to control subjects. This indicates a temporal perception deficit in the range of milliseconds in ADHD may impact upon other functions such as perceptual language skills and motor timing and this agrees with Porras-Alonso et al [38] and Azzam and Hassan [39]; they also mentioned that the processing of auditory information in the brainstem was impaired in ADHD children. Galbraith et al [40] suggested the existence of crude attention mechanisms at the level of the auditory brainstem. These mechanisms could serve to enhance auditory encoding by directing processing resources to the appropriate modality, or within the auditory modality to the appropriate ear. On the contrary, Schochat et al [41] found normal ABR with normal wave latencies in ADHD children included in their study, and their study had small sample size which may explain the difference in the results.
As regard ADHD subtypes, we found significant differences among three subtypes of ADHD with the inattentive ADHD type having the most delayed latencies in waves (III and V) and the intervals (I-III and I-V). These findings suggest that the inattentive ADHD subtype has more impairment in auditory processing and brainstem transmission timing than other subtypes. This agrees with Effat et al [42] who concluded that high co-morbidity exists between auditory processing disorder (APD) and ADHD, with the most affected ability being temporal auditory processing. Inattention and cognitive problems were the only clinical variables correlated to the presence of APD. Bamiou et al [43] mentioned that results arising from diagnostic methodologies and overlapping symptomatology of the two conditions may account for the debate as to whether APD and ADHD are single or two distinct but co-morbid developmental disorders. Clinicians can identify a reasonably exclusive set of diagnostic behavioral characteristics for ADHD and APD. However, consistency does not ensure validity of the diagnosis, and APD and the predominantly inattentive subtype of attention deficit disorder may yet be a single developmental disorder. On the other hand, we disagree with Ahmad Ghanizadeh [44] who reported in his study about screening signs of auditory processing problem in ADHD subtypes that auditory process problems is not predominant in ADHD subtypes.
Our study found 37.5% of ADH children had abnormal non-epileptiform activity in the form of diffuse background slowing and 15% with epileptiform activity. Barry et al [45] reported that ADHD group shows elevated levels of slow wave activity in comparison to normal children. Our results are also in agreement with Hemmer et al [46] who carried out a study on 234 children with ADHD and reported that 15% of them had epileptiform activity. Socanski et al [47] found ADHD children having epileptiform activity consisting 5.4%. Richer et al [48] found epileptiform activity in 6.1%. The difference in percentages of abnormalities could be due to different sample sizes between the different studies and different demographic characteristics of the patients in different studies.
In the present study, the epileptiform activity is more prevalent in inattentive ADHD subtype. This is in agreement with Socanski et al [47] who found that the ADHD-I subtype was more common in children with epileptiform activity independent of a history of epilepsy. They mentioned that such a relationship has not previously been reported in children without co-morbid epilepsy, although it has been observed in patients with epilepsy. Laporte et al [49] mentioned that cognitive dysfunction, attention difficulties and/or behavioral problems in ADHD may be related to the presence of interictal epileptiform activity on EEGs. For example, transient cognitive impairment during frequent subclinical epileptiform discharges can affect attention and cognitive function even in the absence of clinical seizure.
In the present work, learning disabilities had strong negative correlation to score of inattentive type of ADHD but not statistically significant and significant negative correlation to ABR waves latency and epileptiform activity in EEG. Khaliq et al [50] reported that ABR abnormalities have been found in children with learning problems. They found a significant increase in latencies of wave II, III, IV and V, and interpeak latency I-V of ABR in poor performers. Kasteleijn-Nolst [51] found that interictal epileptiform discharges have been demonstrated to cause transitory cognitive impairment through a deleterious effect on attention, perception, reaction times, short-term memory and more complex intellectual tasks in epileptic patients. Furthermore, some children present transient behavioral and learning difficulties correlated with epileptiform discharges without clinical epilepsy.
Conclusion
From the present work, we can conclude that learning disabilities are strongly co-morbid with ADHD especially inattentive type and both may had brainstem and cortical processing abnormalities. Further researches with higher number of patients are needed to support these results.
| References | ▴Top |
- Kieling R, Rohde LA. ADHD in children and adults: diagnosis and prognosis. Curr Top Behav Neurosci. 2012;9:1-16.
doi pubmed - World Health Organization, 1993. The ICD-10 classification of mental and behavioural disorders: diagnostic criteria for research. Geneva: WHO.
- American Psychiatric Association, . Diagnostic and Statistical Manual of Mental Disorders (4th edition, Text Revision) (DSM-IV-TR). Washington, DC:American Psychiatric Association.
- Rubia K. ADHD: What have we learned from Neuroimaging? Cutting edge psychiatry in practice (CEPIP). 2012;1:16-19.
- Valera EM, Faraone SV, Murray KE, Seidman LJ. Meta-analysis of structural imaging findings in attention-deficit/hyperactivity disorder. Biol Psychiatry. 2007;61(12):1361-1369.
doi pubmed - Margari L, Buttiglione M, Craig F, Cristella A, de Giambattista C, Matera E, Operto F, et al. Neuropsychopathological comorbidities in learning disorders. BMC Neurol. 2013;13:198.
doi pubmed - Giraud AL, Ramus F. Neurogenetics and auditory processing in developmental dyslexia. Curr Opin Neurobiol. 2013;23(1):37-42.
doi pubmed - Polanczyk G, Rohde LA. Epidemiology of attention-deficit/hyperactivity disorder across the lifespan. Curr Opin Psychiatry. 2007;20(4):386-392.
doi pubmed - Rohde L. Is there a need to reformulate ADHD criteria in future classification systems? Child Adolesc Psychiatric Clin N Am. 2008;17:405-20.
doi pubmed - Bishop DV. Developmental cognitive genetics: how psychology can inform genetics and vice versa. Q J Exp Psychol (Hove). 2006;59(7):1153-1168.
doi pubmed - Riccio CA, Cohen MJ, Garrison T, Smith B. Auditory processing measures: correlation with neuropsychological measures of attention, memory, and behavior. Child Neuropsychol. 2005;11(4):363-372.
doi pubmed - Cook JR, Mausbach T, Burd L, Gascon GG, Slotnick HB, Patterson B, Johnson RD, et al. A preliminary study of the relationship between central auditory processing disorder and attention deficit disorder. J Psychiatry Neurosci. 1993;18(3):130-137.
pubmed - Tillery KL, Katz J, Keller WD. Effects of methylphenidate (Ritalin) on auditory performance in children with attention and auditory processing disorders. J Speech Lang Hear Res. 2000;43(4):893-901.
pubmed - Azzam H, Hassan D. Auditory Brainstem Timing and Cortical Processing in Attention Deficit Hyperactivity Disorder. Current Psychiatry. 2010;17:15-21.
- Jacobson JT. Normative aspects of the pediatric auditory brainstem response. J Otolaryngol Suppl. 1985;14:7-11.
pubmed - Davis SM, Katusic SK, Barbaresi WJ, Killian J, Weaver AL, Ottman R, Wirrell EC. Epilepsy in children with attention-deficit/hyperactivity disorder. Pediatr Neurol. 2010;42(5):325-330.
doi pubmed - Dunn DW, Kronenberger WG. Childhood epilepsy, attention problems, and ADHD: review and practical considerations. Semin Pediatr Neurol. 2005;12(4):222-228.
doi pubmed - Hughes JR, DeLeo AJ, Melyn MA. The electroencephalogram in attention deficit-hyperactivity disorder: emphasis on epileptiform discharges. Epilepsy Behav. 2000;1(4):271-277.
doi pubmed - Holtmann M, Becker K, Kentner-Figura B, Schmidt MH. Increased frequency of rolandic spikes in ADHD children. Epilepsia. 2003;44(9):1241-1244.
doi pubmed - Swanson JM, Kraemer HC, Hinshaw SP, Arnold LE, Conners CK, Abikoff HB, Clevenger W, et al. Clinical relevance of the primary findings of the MTA: success rates based on severity of ADHD and ODD symptoms at the end of treatment. J Am Acad Child Adolesc Psychiatry. 2001;40(2):168-179.
doi pubmed - Bussing R, Fernandez M, Harwood M, Wei H, Garvan CW, Eyberg SM, Swanson JM. Parent and teacher SNAP-IV ratings of attention deficit hyperactivity disorder symptoms: psychometric properties and normative ratings from a school district sample. Assessment. 2008;15(3):317-328.
doi pubmed - Yaser Salem. Modified Myklebust test for diagnosis of learning disability for Jordan primary school pupils. Journal of studies, Jordon University. 1988;15:133-162.
- Cunningham CE, Barkley RA. The role of academic failure in hyperactive behavior. J Learn Disabil. 1978;11(5):274-280.
doi - Rabiner D, Coie JD. Early attention problems and children's reading achievement: a longitudinal investigation. The Conduct Problems Prevention Research Group. J Am Acad Child Adolesc Psychiatry. 2000;39(7):859-867.
doi pubmed - McGee R, Prior M, Willams S, Smart D, Sanson A. The long-term significance of teacher-rated hyperactivity and reading ability in childhood: findings from two longitudinal studies. J Child Psychol Psychiatry. 2002;43(8):1004-1017.
doi pubmed - Dally K. The influence of phonological processing and inattentive behaviour on reading acquisition. J Educ Psychol. 2006;98:420-437.
doi - Willcutt EG, Pennington BF, DeFries JC. Twin study of the etiology of comorbidity between reading disability and attention-deficit/hyperactivity disorder. Am J Med Genet. 2000;96(3):293-301.
doi - Carroll JM, Maughan B, Goodman R, Meltzer H. Literacy difficulties and psychiatric disorders: evidence for comorbidity. J Child Psychol Psychiatry. 2005;46(5):524-532.
doi pubmed - Kain W, Landerl K, Kaufmann L. Comorbidity of ADHD. Monatsschr Kinderheilkd. 2008;8:757-767.
doi - Langberg JM, Vaughn AJ, Brinkman WB, Froehlich T, Epstein JN. Clinical utility of the Vanderbilt ADHD Rating Scale for ruling out comorbid learning disorders. Pediatrics. 2010;126(5):e1033-1038.
doi pubmed - Willcutt EG, Betjemann RS, McGrath LM, Chhabildas NA, Olson RK, DeFries JC, Pennington BF. Etiology and neuropsychology of comorbidity between RD and ADHD: the case for multiple-deficit models. Cortex. 2010;46(10):1345-1361.
doi pubmed - Ebejer JL, Coventry WL, Byrne B, Willcutt EG, Olson RK, Corley R, Samuelsson S. Genetic and environmental influences on inattention, hyperactivity-impulsivity, and reading: kindergarten to grade 2. Sci Stud Read. 2010;14(4):293-316.
doi pubmed - Greven CU, Rijsdijk FV, Asherson P, Plomin R. A longitudinal twin study on the association between ADHD symptoms and reading. J Child Psychol Psychiatry. 2012;53(3):234-242.
doi pubmed - Fawcett AJ, Nicolson RI. From Dyslexia: The role of the cerebellum. In Dyslexia in context: research, policy and practice. Chapter 2. Edited by Reid G, Fawcett AJ. London: Whurr Publishers; 2004.
doi - Gagliano A, Germano E, Calabrese T, Magazu A, Grosso R, Siracusano RM, Cedro C. La comorbidita nella dislessia: studio di un campione di soggetti in eta evolutiva con disturbo di lettura. Dislessia. 2007;4:21-39.
- Stella G, Franceschi S, Savelli E. Disturbi associati nella dislessia evolutiva. Uno studio preliminare. Dislessia. 2009;6:1.
- Carlson CL, Mann M. Attention-deficit/hyperactivity disorder, predominantly inattentive subtype. Child Adolesc Psychiatr Clin N Am. 2000;9(3):499-510, vi.
pubmed - Porras-Alonso E, Fornell-Forcade J, Domingo-Aznar L. PTC y deficit de atencion e hiper activida d infantil. [Auditory brain stem response and child Attention Deficit Hyperactivity Disorder]. Acta Otorrinolaringologica Espanola. 1999;50:567-570.
pubmed - Azzam H, Hassan D. Auditory Brainstem Timing and Cortical Processing in Attention Deficit Hyperactivity Disorder. Current Psychiatry. 2010;17:15-21.
- Galbraith GC, Olfman DM, Huffman TM. Selective attention affects human brain stem frequency-following response. Neuroreport. 2003;14(5):735-738.
doi pubmed - Schochat E, Scheuer CI, Andrade ER. ABR and auditory P300 findings in children with ADHD. Arq Neuropsiquiatr. 2002;60(3-B):742-747.
doi pubmed - Effat S, Tawfik S, Hussein H, Azzam H and El Eraky S. Central auditory processing in attention deficit hyperactivity disorder: an Egyptian Study. Middle East Current Psychiatry. 2011;18:245-252.
doi - Bamiou DE, Musiek FE, Luxon LM. Aetiology and clinical presentations of auditory processing disorders—a review. Arch Dis Child. 2001;85(5):361-365.
doi pubmed - Ahmad Ghanizadeh. Screening signs of auditory processing problem: Does it distinguish attention deficit hyperactivity disorder subtypes in a clinical sample of children. Int J Pediatr Otorhinolarynol. 2008;73:81-87.
doi pubmed - Barry RJ, Clarke AR, Johnstone SJ. A review of electrophysiology in attention-deficit/hyperactivity disorder: I. Qualitative and quantitative electroencephalography. Clin Neurophysiol. 2003;114(2):171-183.
doi - Hemmer SA, Pasternak JF, Zecker SG, Trommer BL. Stimulant therapy and seizure risk in children with ADHD. Pediatr Neurol. 2001;24(2):99-102.
doi - Socanski D, Herigstad A, Thomsen PH, Dag A, Larsen TK. Epileptiform abnormalities in children diagnosed with attention deficit/hyperactivity disorder. Epilepsy Behav. 2010;19(3):483-486.
doi pubmed - Richer LP, Shevell MI, Rosenblatt BR. Epileptiform abnormalities in children with attention-deficit-hyperactivity disorder. Pediatr Neurol. 2002;26(2):125-129.
doi - Laporte N, Sebire G, Gillerot Y, Guerrini R, Ghariani S. Cognitive epilepsy: ADHD related to focal EEG discharges. Pediatr Neurol. 2002;27(4):307-311.
doi - Khaliq F, Alam KK, Vaney N, Singh TB. Sensory, cognitive and motor assessment of children with poor academic performance: an auditory evoked potential study. Indian J Physiol Pharmacol. 2010;54(3):255-264.
pubmed - Kasteleijn-Nolst Trenite DG. Transient cognitive impairment during subclinical epileptiform electroencephalographic discharges. Semin Pediatr Neurol. 1995;2(4):246-253.
doi
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.
