| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website http://www.neurores.org |
Original Article
Volume 3, Number 2, April 2013, pages 56-61
Stimulating Aδ and C-Fibers in the Lower Limb With a 980 nm Diode Laser
Imre P. Krabbenbosa, d, Christiaan F.P. van Swolb, Eric P.A. van Dongena, Selma C. Trompc, Eduard H.J.F. Boezemanc
aDepartment of Anaesthesiology, Intensive Care and Pain Medicine, St. Antonius Hospital Nieuwegein, the Netherlands
bDepartment of Medical Physics, St. Antonius Hospital Nieuwegein, the Netherlands
cDepartment of Clinical Neurophysiology, St. Antonius Hospital Nieuwegein, the Netherlands
dCorresponding author: Imre P. Krabbenbos, Department of Anaesthesiology, Intensive Care and Pain Medicine, St. Antonius Hospital Nieuwegein, Koekoekslaan 1, 3435 CM Nieuwegein, the Netherlands
Manuscript accepted for publication January 31, 2013
Short title: Diode Laser Stimulation of Aδ and C Fibers
doi: https://doi.org/10.4021/jnr177w
| Abstract | ▴Top |
Background: Laser evoked potentials are increasingly used to investigate the integrity of the nociceptive system. Laser heat stimuli can activate Aδ fibers, activation of C-fibers remains difficult. This study attempts to stimulate Aδand C fibers separately with a ‘grid’ to generate respectively late and ultra-late LEPs. A ‘grid’ is a thin aluminum plate used as a spatial filter to stimulate C-fibers. Furthermore, study subjects pressed a button upon detecting a laser stimulus which was used to measure reaction times (RT) following diode laser stimulation.
Methods: Cutaneous heat stimuli were applied at the Th12and L5 dermatome in seventeen volunteers. Conduction velocities (CV) were calculated by measuring latencies of P2 and reaction times (RT).
Results: Stimulation condition Th12no-grid showed a P2late response at 330 ± 47 ms and L5no-grid at 413 ± 53 ms. Mean reaction time during Th12no-grid was 537 ± 146 ms, L5no-grid 784 ± 334 ms, Th12grid 710 ± 195 ms and L5grid 1,391 ± 336 ms. During stimulation block Th12gridand L5grid ultra-late LEPS could not reliably be generated. Median conduction velocities (CV) and their corresponding range were calculated. The median CVRT no grid was 5.8 m/s (range 1.2 - 43.3). The median CVLEP no grid was 13.8 m/s (range 4.7 - 45.4). The median CVRT grid was 1.9 m/s (range 0.8 - 3.7). Ultra-late LEPs could not be generated, although subjects mentioned a long lasting burning pain during Th12grid and L5grid.
Conclusions: This study questions the feasibility of the ‘grid’ to reliably generate C-fiber responses. Pressing a button upon laser stimulus detection seems preferable for identifying C-fiber stimulation in the lower limb, whereas for Aδ nociceptive pathways laser evoked potentials might be of use.
Keywords: Evoked potentials; Laser; C-fibers; Conduction velocities
| Introduction | ▴Top |
Laser evoked potentials are increasingly used for investigating the functional integrity of the nociceptive system [1, 2]. A synchronized activation of cutaneous nociceptors by laser stimuli allows the recording of time-locked neural responses [3]. Several studies showed abnormal LEPs in patients suffering peripheral, radicular, spinal, midbrain and supraspinal neuraxis lesions [1]. To diagnose patients more specifically regarding different nerve diseases, it is essential to investigate the Aδ and C-system separately and in relation to each other. Thinly myelinated (Aδ) and unmyelinated (C) nociceptors have distinct electrophysiological properties. The LEPs related to Aδ-fibers and C-fiber activations are characterized by different latencies reflecting different conduction velocities (CV) of these fibers in the peripheral nerves and spinal cord [4]. Differences in heat activation threshold and epidermal distribution density have been used to activate C-fibers separately [5, 6]. Furthermore, when nerve compression was used to selectively block Aδ-fibers ‘ultra-late’ LEPS were isolated [7]. The use of a special grid seems to be the most feasible method to generate a C-fiber related brain potential [8]. A grid is a thin aluminium plate used as a spatial filter to selectively stimulate C-fibers. The aim of this study was to calculate conduction velocities of peripheral Aδ and C fibers in healthy volunteers and to test the feasibility of a grid for stimulating C-fibers. In order to calculate conduction velocities of peripheral nociceptive fibers we measured latencies of LEP components. Furthermore, study subjects pressed a button upon the detection of a Aδ- or C-fiber mediated heat stimulus, in order to calculate reaction times and conduction velocities [9, 10].
| Materials and Methods | ▴Top |
Subjects
Experiments were performed on a group of 17 healthy volunteers (11 men and 6 women) aged 23 to 64 years (mean ± SD: 40.9 ± 14.0). None of the volunteers were taking medications or reported a (neurological) disease that might affect pain perception. Our research group has experience with the application of laser stimuli and interpreting evoked brain potentials [11]. Verbal informed consent was obtained before entering the study and all participants were free to withdraw at any time. Participation in this study was in accordance with the local Ethics Committee. Participants were positioned on a comfortable bed in a quiet room kept at 21 - 23 °C, while the light was dimmed. Skin temperature of the foot was measured before and after the experiment. When the temperature was lower than 30 °C, a heat lamp was used during the experiment to keep the feet warm. Participants were asked to relax but focus on the sensation of the laser stimulus, to keep their eyes open, gazing slightly upwards at a fixed point on the wall. Both the participants and experimenters wore protective laser goggles during the stimulations.
Laser
Cutaneous stimuli were applied with a 980 nm diode laser (Biolitec, Ceram Optec, Germany). The skin was blackened with East Indian ink (Pelikan, Hannover, Germany) to reduce superficial reflectance of the skin, to increase absorption of the laser light and to rule out bias by difference in skin pigmentation [12]. The laser onset was triggered manually and in silence. To stimulate the Aδfibers, a stimulus duration of 20 ms, stimulation power of 2.5 W and a spot size hand piece (Biolitec, Ceramoptec, Germany) was used. The intensity of the stimulus was approximately 10.2 mJ/mm2. The location between subsequent stimuli was slightly altered to minimize habituation, nociceptor sensitization and skin irritation. Interstimulus duration randomly varied between 6 and 14 seconds. Laser stimulation with a special grid was used to selectively activate C-fibers. This grid was a 0.1 mm thin titanium plate, measuring 40 mm in length and 60 mm in width. In a 25 mm × 25 mm square of this plate, 26 parallel lines were drawn every 1 mm, producing 26 × 26 intersections. A total of 676 (26 × 26) tiny holes were drilled at these intersections, each with a diameter of 0.4 mm (0.125 mm2) [8]. This spatial filter was placed close to the skin to reduce the effect of diffraction. The laser beam was directed to the skin using the 2.5 mm diameter optic hand piece. The array of holes allowed a 2.5 mm laser beam to pass through 4 to 6 holes depending on placement of the hand piece. Stimulus duration was 20 ms and stimulation power of 4 W. The stimulus energy was approximately 8.2 mJ (4 holes) to 12.2 mJ (6 holes). The stimulation sites were the dorsum of the right foot (L5dermatome) and the 12th thoracic vertebra (Th12dermatome).
Stimulation protocol
Four different stimulation conditions were used. Session 1 included stimulation on the dorsum of the right foot with a grid (L5grid) and stimulation on the dorsum of the right foot without a grid (L5no-grid). Session 2 included stimulation on the back with a grid (Th12grid) and stimulation on the back without a grid (Th12no-grid). Every stimulation block, comprising 10 laser stimuli, was followed by a short break of 30 seconds. Each stimulation condition comprises three stimulation blocks (30 laser stimuli in total). Before switching the stimulation location to Th12, there was a twenty minute break (Fig. 1).
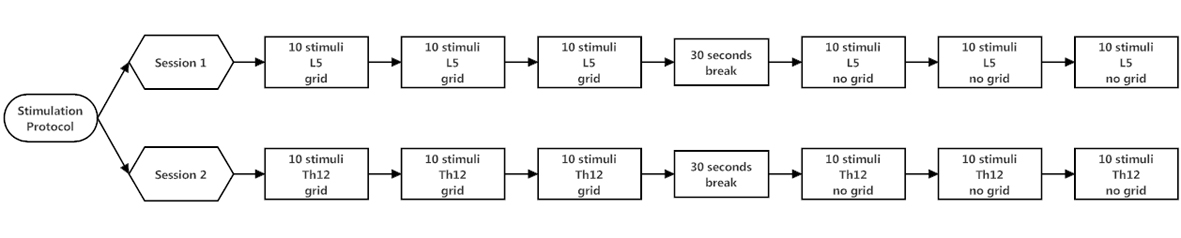 Click for large image | Figure 1. Stimulation protocol. Each stimulation block comprises ten cutaneous laser stimuli, followed by a short break of 30 seconds. First, three stimulation blocks were applied at the dorsum of the right foot (L5 dermatoma) with grid. Three stimulation blocks were applied at the same location without the use of a grid. After a 20 minute break, three stimulation blocks were applied at the 12th thoracic vertebra (Th12 dermatoma) with grid. Subsequently followed by three stimulation blocks without grid at the same location. |
Recording and processing
Evoked potential (EP) recordings were made using silver disc electrodes attached to the scalp according to the International 10-20 system. Exploring electrodes Pz, Cz, C3 and C4 from the vertex were linked to reference electrodes at both earlobes (A1, A2). Impedance was kept below 5 kΩ. EOG was recorded for eye movement artifact filtering. Recordings showing blinks, eye movements or any other artifacts were manually deleted followed by off-line response averaging and analysis. EP signals were sampled at 500 Hz after band-pass filtering (0.2 - 30 Hz). EP epochs were recorded for 2,000 ms with no pre-stimulation delay. Medelec Synergy (Viasys Healthcare, UK) was used to store single sweeps.
Conduction velocities
The participants were instructed to press a button with the index finger of the dominant hand when they perceived a painful sensation [9, 10]. Mean reaction times (RT) were measured over each stimulation block (10 stimuli). The conduction distance (CD) between the two stimuli sites (L5and Th12) was measured from the dorsum of the right foot to the 12th thoracic vertebra along the leg and buttock. The conduction velocities (m/s) of the peripheral nociceptive nerves in the leg were calculated using the differences in peak latencies (P2) and differences in reaction times.
Statistical analysis
The calculated conduction velocities of CVRT no grid andCVLEP no grid showed a non-normal distribution. Therefore, Wilcoxon signed-rank test was used to compare CVRT no grid with CVLEP no grid. Statistical significance was defined as P < 0.05.
| Results | ▴Top |
Seventeen healthy volunteers participated in the study. One participant was excluded because he was not able to feel the laser stimulations.
Grand averages and latencies of LEP signals
For every volunteer, three sets of ten averaged single sweeps were recorded (Fig. 1). Three participants were excluded for the LEP measurements, but included for the reaction time measurements. The LEP signals of these participants were contaminated by eye signals or muscle artifacts and during the experiment it was not possible to control these artifacts. In all other participants reliable LEP signals were generated during stimulation block Th12no-grid and L5no-grid. In Figure 2 and 3 grand averages of LEP recordings are presented. Mean latency of P2 for stimulation condition Th12no-grid and L5no-grid were 330± 47ms and 413 ± 53ms respectively (Table 1). During laser stimulation with the use of a grid (Th12grid and L5grid), no reliable C-fiber-mediated ultra-late LEPs were generated. All study subjects mentioned they experienced a longer lasting burning pain. Sensations were clearly different compared to Aδ stimulation and likely to correspond to C-fiber mediated pain. However, no cortical ultra-late LEPs were recorded.
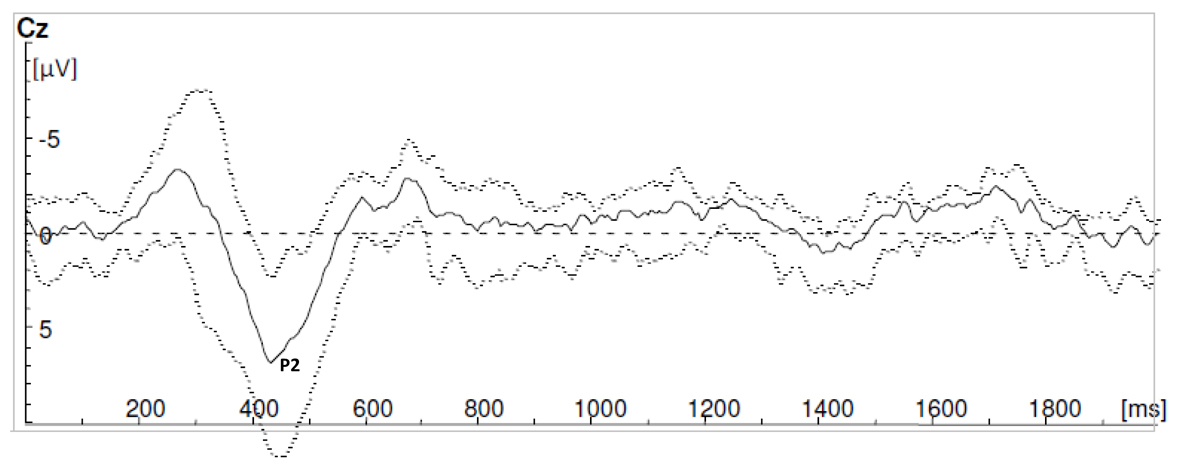 Click for large image | Figure 2. Grand averages of laser evoked potential recordings during stimulation block L5no-grid. |
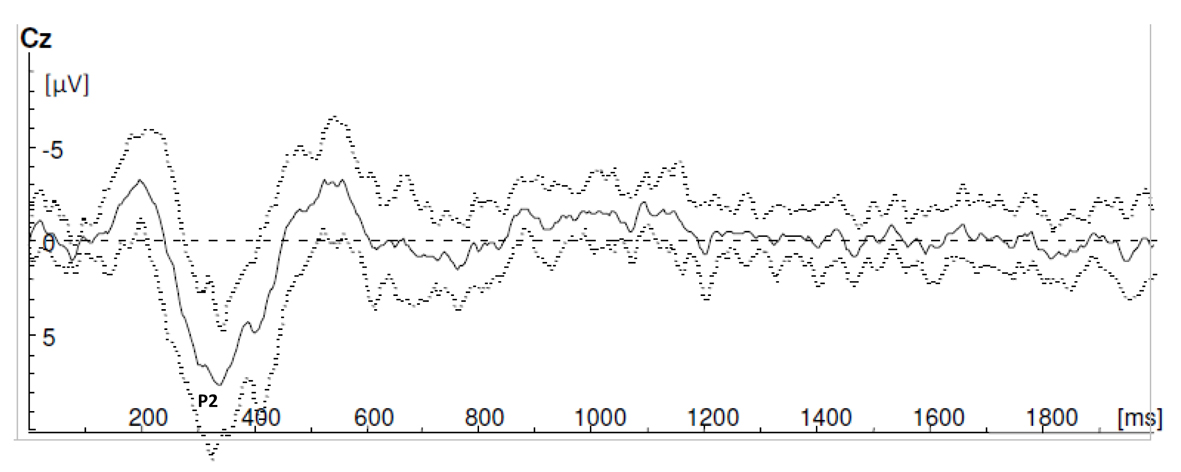 Click for large image | Figure 3. Grand averages of laser evoked potential recordings during stimulation block Th12no-grid. |
 Click to view | Table 1. Laser-Evoked Potentials Peak Latencies (P2) and Reaction Times (RT) |
Reaction times
The mean reaction time for condition Th12no-grid and L5no-grid was 537 ± 146 ms and 784 ± 334ms respectively. Stimulation condition Th12grid and L5gridshowed mean reaction times of 710 ± 195 ms and 1,391 ±336 ms respectively (Table 1). In two participants, one set of ten averaged reaction time measurements was equal during stimulation condition L5grid and L5no-grid, most likely because of concurrent activation of Aδ fibers during condition L5grid. These measurements were subsequently excluded.
Conduction velocities
The conduction distances (CD) between the dorsum of the foot and the 12th thoracic vertebra (1.23 m ± 0.17) were used to calculate conduction velocities based on reaction times (CVRT) and latency (P2) measurements (CVLEP). The calculated conduction velocities of CVRT no grid and CVLEPno grid showed a non-normal distribution, therefore we present median conduction velocities and their corresponding range (Table 2). The range indicates dispersion of data, as it shows the smallest and greatest observation. The median CVRT no grid was 5.8 m/s (range 1.2 - 43.3). The median CVLEP was 13.8 m/s (range 4.7 - 45.4). Statistical analysis, using the Wilcoxon signed-rank test, showed CVRT no grid was significantly lower than CVLEP no grid (P < 0.05). The median CVRT grid was 1.9 m/s (range 0.8 - 3.7) (Table 2).
 Click to view | Table 2. Conduction Velocities (CV) of Peripheral Aδ Fibers and C Fibers |
| Discussion | ▴Top |
Our study questions the feasibility of the ‘grid method’ for generating an ultra-late LEP response in healthy subjects. In literature the ‘grid method’ is presented as the most eligible technique for stimulating C-fibers and generating corresponding ultra-late LEP responses [4, 6]. However, as our study and former literature demonstrates, the selective activation of C-fiber receptors and recording of the related‘ultra late LEP’ brain potentials remains difficult [13]. Since ultra-late LEPs are not reliably present in all healthy subjects, they have not been used to test the integrity of C-fiber mediated nociceptive pathways in patients. It remains a challenge to find an (alternative) method for reliably generating ultra-late LEPs, since it might provide a framework for objectively demonstrating the selective loss of unmyelinated nociceptive fibers. In our study, peak detection during stimulation blocks L5grid and Th12grid was not possible. Thus, C-fiber mediated cortical responses, as presented by Obi et al [4], could not be reproduced in this study because of the low peak-to-peak amplitude. The low peak-to-peak amplitude could have been improved to minimize the distance between the stimulation and recording, because the peak-latency jitter decreases and subsequently peak detection improves [2]. During stimulation with a grid, cutaneous heat stimulation area varied as the laser beam was passing though 4 (0.5 mm2) to 6 (0.76 mm2) holes. The stimulation intensity was approximately between 8.15 - 12.2 mJ/mm2. Former studies used a stimulus intensity of 8.5 ± 1.8 mJ/mm2 for C-fiber activation [14]. We find it remarkable that during stimulation block Th12grid and L5grid all subjects experienced a burning, long lasting sensation corresponding to C-fiber mediated ‘second’ pain. Strikingly, these clearly C-fiber mediated sensations were not subsequently followed by reliable generated ultra-late LEPs. Nonetheless, the measured reaction times clearly correspond to expected values, indicating C-fibers were stimulated. According to the ‘first come first serve hypothesis’, Aδ-fiber stimulation needs to be avoided in order to observe C-fiber mediated ‘ultra-late LEPs [15]. During stimulation block Th12grid and L5grid we did not observe an Aδ-fiber mediated cortical response. Another factor that needs to be discussed, is the laser beam diameter in relation to the density distribution of Aδ- and C-fiber terminals. Former studies assumed the spatial distribution of Aδ- and C-fiber terminals has a Poisson distribution with a average occurrence of kD = 0.5/mm2and kC = 5/mm2 [3]. In our experiment the laser stimulus covers ± 5 holes corresponding to a total stimulation area of 0.625 mm2. The probability of no Aδ-fiber activation P(NAδ(A) = 0) = 0.73 in our experiment. Ideally, the probability of no Aδ-fiber activation is ≥ 0.9. Our failure to record ultra-late LEPs might partially be explained by a suboptimal amount of isolated C-fiber responses to extract ultra-late LEPs by averaging due to concurrent Aδ-fiber activation. Calculated median conduction velocities of Aδfibers (CVRT no grid) were 5.8 m/s (range 1.2 - 43.3) and (CVLEP no grid) 13.8 m/s (range 4.7 - 45.4). The difference between the median CVRT and CVLEP no grid can be explained by concurrent activation of C-fibers during laser stimulation without grid. Although no ultra-late LEPs were visible during EP registration, study subjects might have responded to C-fiber stimulation by pressing the button. During C-fiber stimulation (CVRT grid) calculated median conduction velocities were 1.9 m/s (range 0.8 - 3.7). Both measured Aδ and C-fiber mediated reaction times and conduction velocities equal values described in former literature [4, 16]. However, the variability in the measurements with the diode laser was greater than results presented by Tran et al and Obi et al, using a CO2 laser [4]. The wider range of age (23 to 64 years) in our research group could also be an explanation, as age can influence latency time [2]. Measuring the reaction time with a button is a method to objectivate if the pain stimulus was perceived. Moreover, it enables discrimination between Aδand C-fibers activation. Furthermore, it provides an alternative method to calculate conduction time and conduction velocity. It had been shown that pressing a button (a so-called motor task) upon perceiving a stimulus does not affect the latency of the P2late of late LEP’s as compared to the situation without the motor task [9, 10].Thus, in this study we did not succeed to reliably generate C-fiber cortical responses with the use of a grid. However, the grid seems feasible for stimulating C-fibers, as study participants mentioned a burning pain during stimulation block L5grid and Th12grid. With the help of a button we calculated conduction velocities of Aδ and C-fibers which approaches former literature. This study questions the feasibility of the ‘grid method’ for reliably generating ultra-late LEP fiber responses. Since pressing a button upon laser stimulus detection is simple, reliable and user-friendly it might be in particular preferable for assessing the integrity of C-fiber nociceptive pathways in the lower limb, whereas for Aδ nociceptive pathways laser evoked potentials might be of use.
Conflicts of Interest
The authors declare that no funding was received for this research.
| References | ▴Top |
- Treede RD, Lorenz J, Baumgartner U. Clinical usefulness of laser-evoked potentials. Neurophysiol Clin. 2003;33(6):303-314.
doi pubmed - Bromm B, Lorenz J. Neurophysiological evaluation of pain. Electroencephalogr Clin Neurophysiol. 1998;107(4):227-253.
doi - Plaghki L, Mouraux A. How do we selectively activate skin nociceptors with a high power infrared laser? Physiology and biophysics of laser stimulation. Neurophysiol Clin. 2003;33(6):269-277.
doi - Obi T, Takatsu M, Yamazaki K, Kuroda R, Terada T, Mizoguchi K. Conduction velocities of Adelta-fibers and C-fibers in human peripheral nerves and spinal cord after CO2 laser stimulation. J Clin Neurophysiol. 2007;24(3):294-297.
doi pubmed - Magerl W, Ali Z, Ellrich J, Meyer RA, Treede RD. C- and A delta-fiber components of heat-evoked cerebral potentials in healthy human subjects. Pain. 1999;82(2):127-137.
doi - Bragard D, Chen AC, Plaghki L. Direct isolation of ultra-late (C-fibre) evoked brain potentials by CO2 laser stimulation of tiny cutaneous surface areas in man. Neurosci Lett. 1996;209(2):81-84.
doi - Bromm B, Neitzel H, Tecklenburg A, Treede RD. Evoked cerebral potential correlates of C-fibre activity in man. Neurosci Lett. 1983;43(1):109-114.
doi - Tran TD, Lam K, Hoshiyama M, Kakigi R. A new method for measuring the conduction velocities of Abeta-, Adelta- and C-fibers following electric and CO(2) laser stimulation in humans. Neurosci Lett. 2001;301(3):187-190.
doi - Siedenberg R, Treede RD. Laser-evoked potentials: exogenous and endogenous components. Electroencephalogr Clin Neurophysiol. 1996;100(3):240-249.
doi - Opsommer E, Masquelier E, Plaghki L. Determination of nerve conduction velocity of C-fibres in humans from thermal thresholds to contact heat (thermode) and from evoked brain potentials to radiant heat (CO2 laser). Neurophysiol Clin. 1999;29(5):411-422.
doi - Krabbenbos IP, Brandsma D, van Swol CF, Boezeman EH, Tromp SC, Nijhuis HJ, van Dongen EP. Inhibition of cortical laser-evoked potentials by transcutaneous electrical nerve stimulation. Neuromodulation. 2009;12(2):141-145.
doi pubmed - Arendt-Nielsen L, Bjerring P. Reaction times to painless and painful CO2 and argon laser stimulation. Eur J Appl Physiol Occup Physiol. 1988;58(3):266-273.
doi - Cruccu G, Pennisi E, Truini A, Iannetti GD, Romaniello A, Le Pera D, De Armas L, et al. Unmyelinated trigeminal pathways as assessed by laser stimuli in humans. Brain. 2003;126(Pt 10):2246-2256.
doi pubmed - Mouraux A, Plaghki L. Are laser-evoked brain potentials modulated by attending to first or second pain? Pain. 2007;129(3):321-331.
doi pubmed - Garcia-Larrea L. Somatosensory volleys and cortical evoked potentials: 'first come, first served'? Pain. 2004;112(1-2):5-7.
doi pubmed - Tran TD, Inui K, Hoshiyama M, Lam K, Kakigi R. Conduction velocity of the spinothalamic tract following CO2 laser stimulation of C-fibers in humans. Pain. 2002;95(1-2):125-131.
doi
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.







